Alzamend Neuro, Inc. (ALZN): Price and Financial Metrics
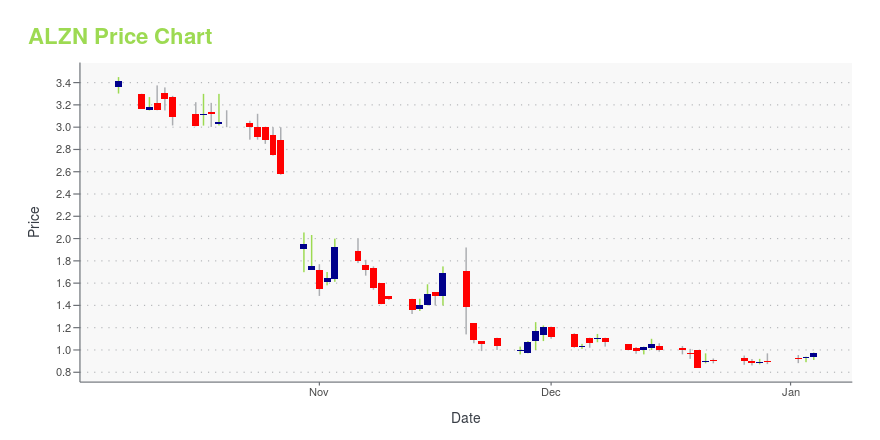
ALZN Price/Volume Stats
| Current price | $0.69 | 52-week high | $11.91 |
| Prev. close | $0.72 | 52-week low | $0.68 |
| Day low | $0.68 | Volume | 27,600 |
| Day high | $0.72 | Avg. volume | 53,801 |
| 50-day MA | $0.94 | Dividend yield | N/A |
| 200-day MA | $2.56 | Market Cap | 4.75M |
ALZN Stock Price Chart Interactive Chart >
Alzamend Neuro, Inc. (ALZN) Company Bio
Alzamend Neuro, Inc., a preclinical stage biopharmaceutical company, focuses on developing products for the treatment of neurodegenerative diseases and psychiatric disorders. The company's lead product candidate is AL001 for the treatment of Alzheimer's and other neurodegenerative diseases and psychiatric disorders. It is also developing AL002, a cell-based therapeutic vaccine, which seeks to restore the ability of the patient's immunological system to Alzheimer's. The company was incorporated in 2016 and is headquartered in Tampa, Florida.
Latest ALZN News From Around the Web
Below are the latest news stories about ALZAMEND NEURO INC that investors may wish to consider to help them evaluate ALZN as an investment opportunity.
EXCLUSIVE: Alzamend Neuro Plans To Kickstart Stress Disorder Study With Next-Gen Lithium Product Next YearAlzamend Neuro Inc (NASDAQ: ALZN) received a "Study May Proceed" letter from the FDA for the initiation of study AL001-PTSD01, a Phase 2A study of AL001 for post-traumatic stress disorder (PTSD). "Although lithium does not have an FDA-approved indication for PTSD, it has been prescribed off-label for this purpose for decades," said Stephan Jackman, Chief Executive Officer of Alzamend. "If we can develop a next-generation lithium product (AL001) that would not routinely require therapeutic drug m |
Alzamend Neuro Receives FDA "Study May Proceed" Notification for a Phase IIA Clinical Trial of AL001, a Next‑Generation Lithium Therapeutic Drug Candidate, in Post-Traumatic Stress Disorder PatientsATLANTA, December 11, 2023--Alzamend Neuro Receives FDA "Study May Proceed" Notice for Phase IIA Clinical Trial of AL001, a Next-Generation Lithium Therapeutic Drug to Treat PTSD |
Today’s Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on TuesdayIt's time to start Tuesday trading with a breakdown of the biggest pre-market stock movers worth keeping an eye on this morning! |
Alzamend Neuro Receives FDA "Study May Proceed" Notification for a Phase IIA Clinical Trial of AL001, a Next‑Generation Lithium Therapeutic Drug Candidate, in Major Depressive Disorder PatientsATLANTA, November 20, 2023--Alzamend Gets FDA "Study May Proceed" Notice for Phase IIA Clinical Trial of AL001 a Next-Generation Lithium Therapeutic for Major Depressive Disorder |
Alzamend Neuro Regains Compliance with Nasdaq’s Minimum Bid Price RequirementATLANTA, November 16, 2023--Alzamend Neuro Regains Compliance with Nasdaq’s Minimum Bid Price Requirement |
ALZN Price Returns
| 1-mo | -33.65% |
| 3-mo | -22.21% |
| 6-mo | -77.13% |
| 1-year | -92.20% |
| 3-year | N/A |
| 5-year | N/A |
| YTD | -22.47% |
| 2023 | -89.50% |
| 2022 | -70.27% |
| 2021 | N/A |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...