ASLAN Pharmaceuticals Limited (ASLN): Price and Financial Metrics
ASLN Price/Volume Stats
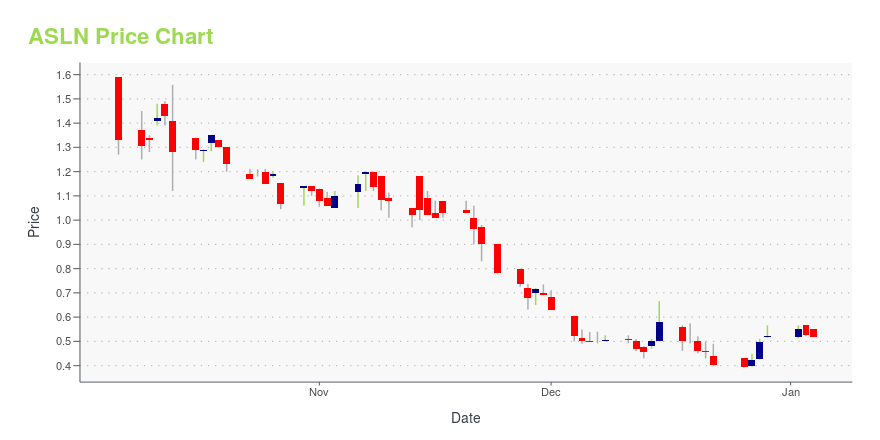
| Current price | $0.47 | 52-week high | $4.69 |
| Prev. close | $0.46 | 52-week low | $0.39 |
| Day low | $0.45 | Volume | 1,517,000 |
| Day high | $0.52 | Avg. volume | 1,001,597 |
| 50-day MA | $0.61 | Dividend yield | N/A |
| 200-day MA | $1.19 | Market Cap | 7.67M |
ASLN Stock Price Chart Interactive Chart >
ASLAN Pharmaceuticals Limited (ASLN) Company Bio
ASLAN Pharmaceuticals Pte. Ltd., a biotechnology company, develops immuno-oncology agents and targeted therapies. It offers ASLAN001, a pan-HER inhibitor for solid tumours; and ASLAN002, a cMET inhibitor for solid tumours. The company also inlicenses preclinical and early clinical/development compounds and candidate drugs from pharmaceutical companies focusing on oncology, respiratory, and inflammation diseases; gets proof of concept (PoC) for the partnered compounds in Asia; and outlicenses PoC compounds back to partner for development and launch. It sells its products in Asia and internationally. ASLAN Pharmaceuticals Pte. Ltd. was founded in 2010 and is based in Singapore.
Latest ASLN News From Around the Web
Below are the latest news stories about ASLAN PHARMACEUTICALS LTD that investors may wish to consider to help them evaluate ASLN as an investment opportunity.
ASLAN Pharmaceuticals Provides Year-End Update on Its Eblasakimab and Farudodstat ProgramsFollowing the successful TREK-AD Phase 2b study of eblasakimab in moderate-to-severe atopic dermatitis, process is underway to identify potential partners. Recruitment in TREK-DX, studying eblasakimab in dupilumab-experienced patients, using updated criteria based on findings from TREK-AD, has commenced at US sites, with additional sites in Europe expected to open in the first half of 2024.Review of blinded safety data emerging from FAST-AA study of farudodstat in alopecia areata shows no emergi |
ASLAN Pharmaceuticals to Participate in the 35th Annual Piper Sandler Healthcare ConferenceSAN MATEO, Calif. and SINGAPORE, Nov. 20, 2023 (GLOBE NEWSWIRE) -- ASLAN Pharmaceuticals (Nasdaq:ASLN), a clinical-stage, immunology focused biopharmaceutical company developing innovative treatments to transform the lives of patients, today announced Dr Carl Firth, CEO, will participate in a fireside discussion at the Piper Sandler Healthcare Conference. The conference will be held from November 28 to 30, 2023, in-person at the Lotte New York Palace Hotel, in New York. 35TH Annual Piper Sandler |
ASLAN Pharmaceuticals Presents New Data on Eblasakimab in Atopic Dermatitis and COPD Translational Models at the 7th Dermatology Drug Development SummitHead-to-head study between eblasakimab and dupilumab in skin biopsies from atopic dermatitis (AD) patients confirm eblasakimab’s differentiated effects of targeting IL-13R vs IL-4RUsing an established COPD model of human lung slices, new data demonstrate eblasakimab’s potential to reduce airway constriction and enhance dilationResults demonstrate for the first time the potential utility of eblasakimab in other indications beyond AD SAN MATEO, Calif. and SINGAPORE, Nov. 03, 2023 (GLOBE NEWSWIRE) |
ASLAN Pharmaceuticals Reports Third Quarter 2023 Financial Results and Provides Corporate UpdatePositive topline data reported in July from TREK-AD, a Phase 2b study of eblasakimab in moderate-to-severe atopic dermatitis, met the primary endpoint across three dosing arms; the study established eblasakimab’s potential to deliver a monthly dosing regimen from initiation with a competitive efficacy profile. Preparations underway for the advancement of eblasakimab into Phase 3 clinical development in 2024.Topline interim data from the FAST-AA (FArudodstat STudy in Alopecia Areata) Phase 2a stu |
ASLAN Pharmaceuticals to Co-Host KOL Panel Discussion Today on Changes in the Clinical Trial and Treatment Landscape for Atopic DermatitisDiscussion featuring KOLs Dr Jonathan Silverberg, Dr April W. Armstrong, and a leading CRO will explore “The Changing Face of Atopic Dermatitis: How the Clinical Trial and Treatment Landscape Has Changed in the Seven Years Following Dupilumab’s Introduction”ASLAN management will present additional analyses from the TREK-AD study and new market research from prescriber and patient surveys on the AD treatment landscape Register here to attend the webcast event today at 11:00am ET SAN MATEO, Calif. |
ASLN Price Returns
| 1-mo | -28.06% |
| 3-mo | -18.26% |
| 6-mo | -55.87% |
| 1-year | -88.76% |
| 3-year | -97.06% |
| 5-year | -97.63% |
| YTD | -9.98% |
| 2023 | -70.99% |
| 2022 | -67.86% |
| 2021 | -38.80% |
| 2020 | -9.85% |
| 2019 | -43.61% |

Loading social stream, please wait...