Brainstorm Cell Therapeutics Inc. (BCLI): Price and Financial Metrics
BCLI Price/Volume Stats
| Current price | $0.54 | 52-week high | $3.34 |
| Prev. close | $0.53 | 52-week low | $0.13 |
| Day low | $0.52 | Volume | 91,981 |
| Day high | $0.55 | Avg. volume | 985,752 |
| 50-day MA | $0.47 | Dividend yield | N/A |
| 200-day MA | $0.66 | Market Cap | 36.90M |
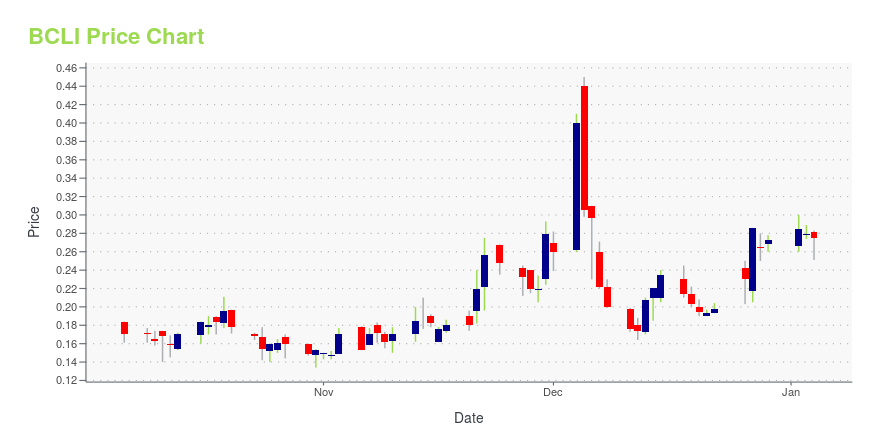
BCLI Stock Price Chart Interactive Chart >
Brainstorm Cell Therapeutics Inc. (BCLI) Company Bio
Brainstorm Cell Therapeutics Inc., a biotechnology company, develops adult stem cell therapies for neurodegenerative disorders. The company was founded in 2000 and is based in Hackensack, New Jersey.
Latest BCLI News From Around the Web
Below are the latest news stories about BRAINSTORM CELL THERAPEUTICS INC that investors may wish to consider to help them evaluate BCLI as an investment opportunity.
BrainStorm Granted Patents for Allogeneic Exosome Platform-Product in Neurological Disorder TreatmentsBrainstorm Cell Therapeutics Inc. (NASDAQ: BCLI), a leading developer of adult stem cell therapeutics for neurodegenerative diseases, today provided an update on the grant and allowance of three patent applications for NurOwn® and NurOwn-Exosomes. |
BrainStorm Issues 2023 Letter to ShareholdersBrainStorm Cell Therapeutics Inc. (NASDAQ: BCLI), a leading developer of adult stem cell therapeutics for neurodegenerative diseases, today issued a Letter to shareholders. |
BCLI: FDA Meeting Provides Clear Path for Planned Phase 3b Trial of NurOwnBy David Bautz, PhD NASDAQ:BCLI READ THE FULL BCLI RESEARCH REPORT Business Update Clear Path Forward for Phase 3b Trial of NurOwn On December 7, 2023, BrainStorm Cell Therapeutics, Inc. (NASDAQ:BCLI) announced the company attended a meeting with the U.S. Food and Drug Administration (FDA) regarding the planned Phase 3b trial for NurOwn in amyotrophic lateral sclerosis (ALS). The company is going |
BrainStorm Cell Therapeutics Announces Outcome of FDA Meeting on NurOwn® in ALSBrainStorm Cell Therapeutics Inc. (NASDAQ: BCLI), a leading developer of adult stem cell therapeutics for neurodegenerative diseases, today announced the completion of a productive meeting with the U.S. Food and Drug Administration (FDA) to discuss NurOwn®, its investigational treatment for amyotrophic lateral sclerosis (ALS). |
BrainStorm Cell Therapeutics Announces In-Person Meeting with the FDA to Discuss Confirmatory Phase 3 Trial for NurOwn® in ALSBrainStorm Cell Therapeutics Inc. (NASDAQ: BCLI), a leading developer of adult stem cell therapeutics for neurodegenerative diseases, today announced that the US Food & Drug Administration (US FDA) has granted the company a meeting to discuss the regulatory path forward for NurOwn® in amyotrophic lateral sclerosis (ALS). The meeting is scheduled to take place on December 6, 2023. Brainstorm will discuss plans for a Special Protocol Assessment (SPA) with the FDA to agree on the overall protocol d |
BCLI Price Returns
| 1-mo | -17.56% |
| 3-mo | 74.08% |
| 6-mo | 237.50% |
| 1-year | -80.58% |
| 3-year | -84.39% |
| 5-year | -86.53% |
| YTD | 97.80% |
| 2023 | -83.35% |
| 2022 | -59.00% |
| 2021 | -11.60% |
| 2020 | 5.72% |
| 2019 | 20.56% |

Loading social stream, please wait...