Black Diamond Therapeutics Inc. (BDTX): Price and Financial Metrics
BDTX Price/Volume Stats
| Current price | $5.02 | 52-week high | $6.85 |
| Prev. close | $5.26 | 52-week low | $1.43 |
| Day low | $4.96 | Volume | 512,700 |
| Day high | $5.47 | Avg. volume | 587,536 |
| 50-day MA | $4.86 | Dividend yield | N/A |
| 200-day MA | $3.48 | Market Cap | 259.62M |
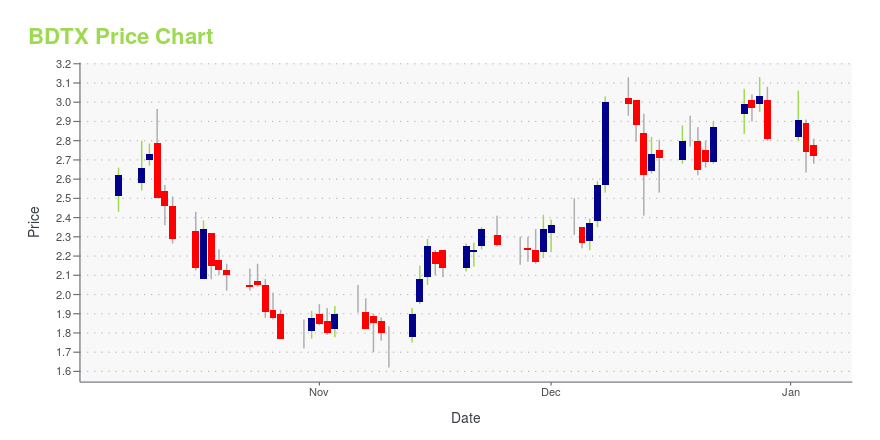
BDTX Stock Price Chart Interactive Chart >
Black Diamond Therapeutics Inc. (BDTX) Company Bio
Black Diamond Therapeutics, Inc. engages in the biotechnology company that discovers and develops therapeutic agents to target unique oncogenic protein-isoforms. The company was founded by Dr. David M. Epstein and Dr. Elizabeth Buck in 2014 and is headquartered in Cambridge, MA.
Latest BDTX News From Around the Web
Below are the latest news stories about BLACK DIAMOND THERAPEUTICS INC that investors may wish to consider to help them evaluate BDTX as an investment opportunity.
Black Diamond Therapeutics Announces Topline Results from Phase 1 Dose Escalation Trial of BDTX-1535 in Patients with Recurrent GBMInitial results show promising clinical activity in heavily pretreated patients 22 patients evaluable for efficacy: 3 patients on therapy longer than 10 months, 1 patient longer than 6 months, and 5 patients longer than 4 months 19 patients with measurable disease by RANO criteria: 1 patient with confirmed partial response and 8 patients with stable disease BDTX-1535 generally well tolerated, consistent with prior disclosures and no new safety signals observed “Window of opportunity” trial enrol |
With 58% ownership, Black Diamond Therapeutics, Inc. (NASDAQ:BDTX) boasts of strong institutional backingKey Insights Significantly high institutional ownership implies Black Diamond Therapeutics' stock price is sensitive to... |
Black Diamond Therapeutics to Participate in Upcoming Investor ConferencesCAMBRIDGE, Mass. and NEW YORK, Nov. 07, 2023 (GLOBE NEWSWIRE) -- Black Diamond Therapeutics, Inc. (Nasdaq: BDTX), a clinical-stage oncology company developing MasterKey therapies that target families of oncogenic mutations in patients with genetically defined cancers, today announced that its President and Chief Executive Officer, Mark A. Velleca, M.D., Ph.D., will participate in fireside chats during two upcoming investor conferences: The Stifel 2023 Healthcare Conference at 2:25pm ET on Tuesda |
Black Diamond Therapeutics Inc (BDTX) Reports Q3 2023 Financial Results and Corporate UpdateCompany's cash position stands at $144.3 million, sufficient to fund operations into H1 2025 |
Black Diamond Therapeutics Reports Third Quarter 2023 Financial Results and Provides Corporate UpdatePresented BDTX-1535 Phase 1 dose escalation data showing durable anti-tumor activity and favorable safety profile in NSCLC patients across heterogeneous EGFR mutations at the October 2023 AACR-NCI-EORTC International Conference on Molecular Targets and Cancer TherapeuticsBDTX-1535 NSCLC expansion cohorts enrolling, initial data expected in 2024BDTX-1535 Phase 1 dose escalation data in GBM expected later this yearDosed first patient in a Phase 1 trial of BDTX-4933, a brain-penetrant RAF inhibitor |
BDTX Price Returns
| 1-mo | -12.54% |
| 3-mo | 79.29% |
| 6-mo | 167.02% |
| 1-year | 217.72% |
| 3-year | -81.18% |
| 5-year | N/A |
| YTD | 78.65% |
| 2023 | 56.11% |
| 2022 | -66.23% |
| 2021 | -83.37% |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...