Oncolytics Biotech Inc. (ONCY): Price and Financial Metrics
ONCY Price/Volume Stats
| Current price | $1.06 | 52-week high | $3.39 |
| Prev. close | $1.03 | 52-week low | $0.88 |
| Day low | $1.03 | Volume | 116,100 |
| Day high | $1.06 | Avg. volume | 381,496 |
| 50-day MA | $1.06 | Dividend yield | N/A |
| 200-day MA | $1.65 | Market Cap | 79.95M |
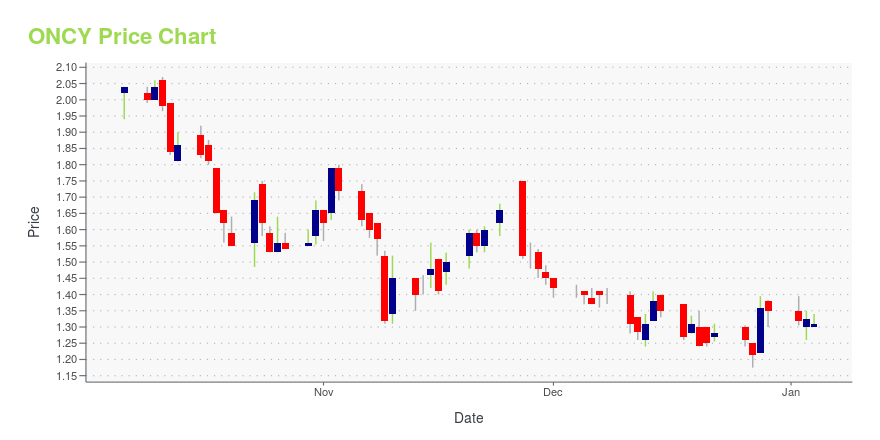
ONCY Stock Price Chart Interactive Chart >
Oncolytics Biotech Inc. (ONCY) Company Bio
Oncolytics Biotech Inc. discovers and develops pharmaceutical products for the treatment of cancers that have not been successfully treated with conventional therapeutics. The Company is focusing on the development of the reovirus known as REOLYSIN as a potential cancer therapeutic.
Latest ONCY News From Around the Web
Below are the latest news stories about ONCOLYTICS BIOTECH INC that investors may wish to consider to help them evaluate ONCY as an investment opportunity.
Oncolytics Biotech® and SOLTI Present Positive Translational Data at SABCSOncolytics Biotech® Inc. (NASDAQ: ONCY) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, today announced the presentation of additional translational data from the AWARE-1 breast cancer window-of-opportunity study conducted in combination with SOLTI-Innovative Cancer Research at The San Antonio Breast Cancer Symposium (SABCS). |
Oncolytics Announces the Anal Cancer Cohort of the GOBLET Phase 1/2 Study of Pelareorep and Atezolizumab Has Met the Success Criteria for EfficacyOncolytics Biotech® Inc. (NASDAQ: ONCY) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, today announced the presentation of positive, interim results from the Phase 1/2 GOBLET study evaluating the combination of pelareorep and atezolizumab... |
Oncolytics Provides Update on Pancreatic Cancer Program for PelareorepOncolytics Biotech® Inc. (NASDAQ: ONCY) (Oncolytics) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, provided an update on the planned program for pelareorep in pancreatic ductal adenocarcinoma (PDAC). |
Oncolytics Biotech Inc. (NASDAQ:ONCY) Q3 2023 Earnings Call TranscriptOncolytics Biotech Inc. (NASDAQ:ONCY) Q3 2023 Earnings Call Transcript November 3, 2023 Oncolytics Biotech Inc. misses on earnings expectations. Reported EPS is $-0.1 EPS, expectations were $-0.07. Operator: Good morning, and welcome to Oncolytics Biotech’s Third Quarter 2023 Conference Call. All participants are now in listen-only mode. There will be a question-and-answer session at the […] |
Oncolytics Biotech® and SOLTI Present Further Positive Pelareorep Translational Data at SITCOncolytics Biotech® Inc. (NASDAQ: ONCY) (TSX: ONC), a clinical-stage immunotherapeutics company focused on oncology, today announced the presentation of a poster that provides further positive translational data from the previously completed AWARE-1 breast cancer window-of-opportunity study, sponsored by SOLTI-Innovative Cancer Research, at the Society for Immunotherapy of Cancer (SITC) 38th Annual Meeting. |
ONCY Price Returns
| 1-mo | 4.95% |
| 3-mo | -12.40% |
| 6-mo | -31.61% |
| 1-year | -10.92% |
| 3-year | -64.90% |
| 5-year | -45.08% |
| YTD | -21.48% |
| 2023 | -17.18% |
| 2022 | 17.27% |
| 2021 | -41.60% |
| 2020 | -50.00% |
| 2019 | 162.98% |

Loading social stream, please wait...