Abeona Therapeutics Inc. (ABEO): Price and Financial Metrics
ABEO Price/Volume Stats
| Current price | $5.22 | 52-week high | $9.01 |
| Prev. close | $5.24 | 52-week low | $3.05 |
| Day low | $5.21 | Volume | 170,200 |
| Day high | $5.32 | Avg. volume | 305,918 |
| 50-day MA | $5.89 | Dividend yield | N/A |
| 200-day MA | $5.48 | Market Cap | 226.92M |
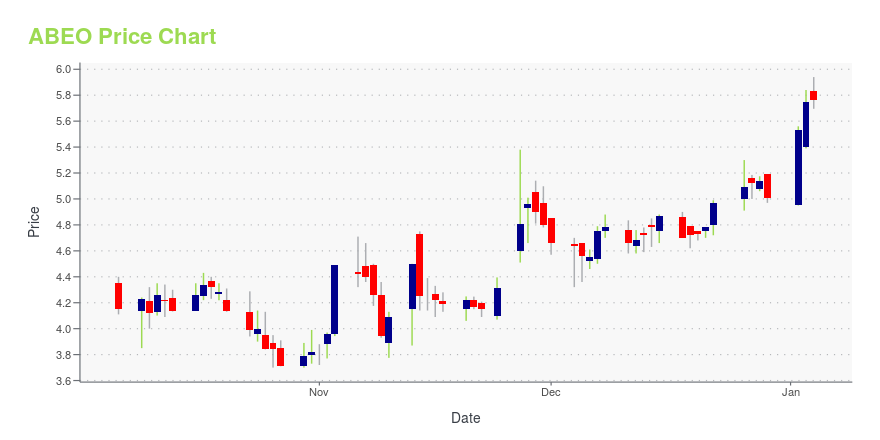
ABEO Stock Price Chart Interactive Chart >
Abeona Therapeutics Inc. (ABEO) Company Bio
Abeona Therapeutics Inc. focuses on developing and delivering gene therapy and plasma-based products for severe and life-threatening rare diseases. The company iwas founded n 1989 and is based in Dallas, Texas.
ABEO Price Returns
| 1-mo | -8.42% |
| 3-mo | -20.55% |
| 6-mo | 3.37% |
| 1-year | 4.40% |
| 3-year | -9.26% |
| 5-year | -92.70% |
| YTD | -6.28% |
| 2024 | 11.18% |
| 2023 | 62.66% |
| 2022 | -63.44% |
| 2021 | -78.54% |
| 2020 | -51.99% |
Continue Researching ABEO
Want to see what other sources are saying about Abeona Therapeutics Inc's financials and stock price? Try the links below:Abeona Therapeutics Inc (ABEO) Stock Price | Nasdaq
Abeona Therapeutics Inc (ABEO) Stock Quote, History and News - Yahoo Finance
Abeona Therapeutics Inc (ABEO) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...