Applied Therapeutics, Inc. (APLT): Price and Financial Metrics
APLT Price/Volume Stats
| Current price | $5.22 | 52-week high | $9.39 |
| Prev. close | $5.20 | 52-week low | $1.18 |
| Day low | $5.16 | Volume | 331,791 |
| Day high | $5.32 | Avg. volume | 1,507,834 |
| 50-day MA | $4.74 | Dividend yield | N/A |
| 200-day MA | $4.06 | Market Cap | 596.47M |
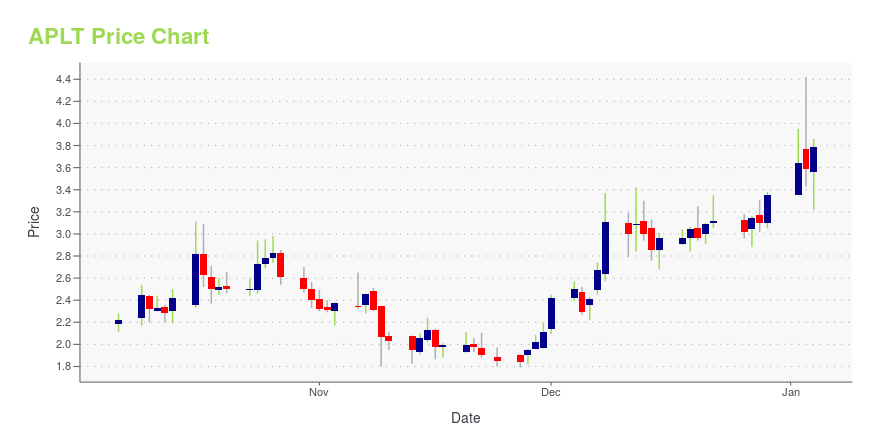
APLT Stock Price Chart Interactive Chart >
Applied Therapeutics, Inc. (APLT) Company Bio
Applied Therapeutics, Inc. is a clinical-stage biopharmaceutical company, which engages in developing a pipeline of novel product candidates against validated molecular targets in indications of high unmet medical need. The company was founded by Shoshana Shendelman on January 20, 2016 and is headquartered in New York, NY
Latest APLT News From Around the Web
Below are the latest news stories about APPLIED THERAPEUTICS INC that investors may wish to consider to help them evaluate APLT as an investment opportunity.
Today’s Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on TuesdayIt's time to dive into the biggest pre-market stock movers as we check out all of the most recent news for Tuesday morning! |
Applied Therapeutics Inc (APLT) Reports Q3 2023 Financial Results Amidst Upcoming Clinical ...Regulatory Submissions and Phase 3 Trials in Focus |
Applied Therapeutics Reports Third Quarter 2023 Financial ResultsRegulatory submissions on track for govorestat (AT-007) for the treatment of Classic Galactosemia to US FDA and EMA in 4Q 2023 Two Upcoming Phase 3 Trial Readouts, with ARISE-HF Trial of AT-001 (caficrestat) in Diabetic Cardiomyopathy on track for data readout in 4Q 2023 and INSPIRE Trial of AT-007 trial in Sorbitol Dehydrogenase (SORD) Deficiency in 1Q 2024 NEW YORK, Nov. 09, 2023 (GLOBE NEWSWIRE) -- Applied Therapeutics, Inc. (Nasdaq: APLT), a clinical-stage biopharmaceutical company developin |
Applied Therapeutics, Inc. (NASDAQ:APLT) is definitely on the radar of institutional investors who own 38% of the companyKey Insights Institutions' substantial holdings in Applied Therapeutics implies that they have significant influence... |
Applied Therapeutics to Present at the UBS Biopharma Conference 2023NEW YORK, Nov. 02, 2023 (GLOBE NEWSWIRE) -- Applied Therapeutics, Inc. (Nasdaq: APLT), a clinical-stage biopharmaceutical company developing a pipeline of novel drug candidates against validated molecular targets in indications of high unmet medical need, today announced that it will present at the UBS Biopharma Conference 2023 on Thursday, November 9, 2023 at 2:00 p.m. ET in Miami, Florida. A live webcast for this event will be accessible on the Events page under the Investor Relations section |
APLT Price Returns
| 1-mo | 17.04% |
| 3-mo | 21.96% |
| 6-mo | 79.38% |
| 1-year | 248.00% |
| 3-year | -70.21% |
| 5-year | -40.34% |
| YTD | 55.82% |
| 2023 | 340.79% |
| 2022 | -91.51% |
| 2021 | -59.34% |
| 2020 | -19.32% |
| 2019 | N/A |

Loading social stream, please wait...