arGEN-X SE ADR (ARGX): Price and Financial Metrics
ARGX Price/Volume Stats
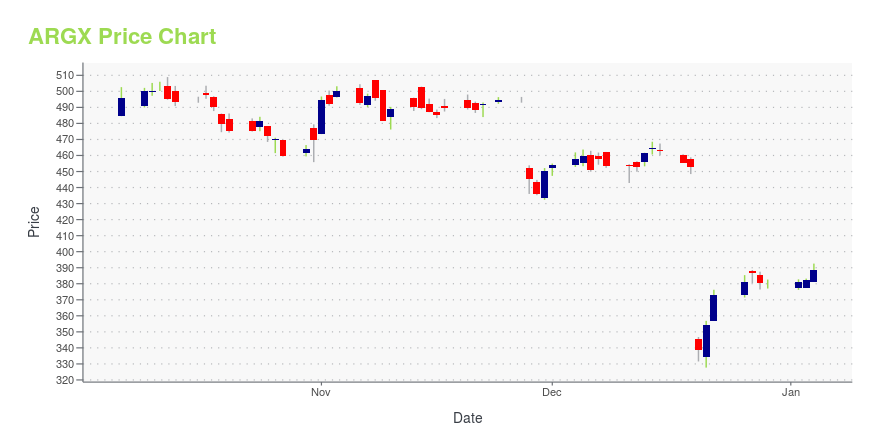
| Current price | $494.46 | 52-week high | $532.59 |
| Prev. close | $486.33 | 52-week low | $327.73 |
| Day low | $473.67 | Volume | 770,000 |
| Day high | $495.50 | Avg. volume | 324,736 |
| 50-day MA | $414.41 | Dividend yield | N/A |
| 200-day MA | $414.61 | Market Cap | 29.39B |
ARGX Stock Price Chart Interactive Chart >
arGEN-X SE ADR (ARGX) Company Bio
argenx (Euronext & Nasdaq: ARGX), a global immunology company committed to improving the lives of people suffering from severe autoimmune diseases and cancers, today announced its 2021 corporate priorities and highlighted recent achievements from its late-stage immunology pipeline driven by its FcRn antagonist, efgartigimod. Additionally, the Company announced interim data from the Phase 2 CULMINATE trial of cusatuzumab in development with Cilag GmbH International, an affiliate of Janssen, and provided financial guidance for 2021. (Source:Wiki)
Latest ARGX News From Around the Web
Below are the latest news stories about ARGENX SE that investors may wish to consider to help them evaluate ARGX as an investment opportunity.
UPDATE 2-European shares end slightly higher ahead of Christmas holidayEuropean stocks closed with a whimper on Friday, as softer-than-expected U.S. inflation data offset losses in sportwear makers and China-exposed stocks ahead of the Christmas holiday weekend. The pan-European STOXX 600 index edged up 0.1% and notched its sixth week of gains in a row - a winning streak that was last seen in December 2022. European markets will be shut on Monday for Christmas. |
Immunovant Scores Much-Needed Win Following Argenx-Led Sell-offImmunovant unveiled promising results for a Graves' disease treatment on Thursday, helping IMVT stock reverse a recent downfall. |
argenx (ARGX) Tanks on Top-Line Data From Pemphigus Studyargenx's (ARGX) phase III ADDRESS study, evaluating efgartigimod subcutaneous for treating pemphigus, fails to meet primary and secondary endpoints. Stock falls. |
Why Is Argenx (ARGX) Stock Down 25% Today?Following a late-stage clinical disappointment, Argenx has been forced back to the drawing board, thus cratering ARGX stock. |
Stock Market Does Sharp About-Face; Dow Breaks Win StreakStock market indexes unexpectedly reversed course in afternoon trading Wednesday, sending the major indexes lower by more than 1%. The Dow Jones Industrial Average couldn't add to its nine-day win streak after reaching all-time highs. |
ARGX Price Returns
| 1-mo | 10.90% |
| 3-mo | 32.63% |
| 6-mo | 29.24% |
| 1-year | -2.87% |
| 3-year | 64.24% |
| 5-year | 246.99% |
| YTD | 29.97% |
| 2023 | 0.42% |
| 2022 | 8.18% |
| 2021 | 19.08% |
| 2020 | 83.21% |
| 2019 | 67.09% |
Continue Researching ARGX
Want to do more research on Argenx Se's stock and its price? Try the links below:Argenx Se (ARGX) Stock Price | Nasdaq
Argenx Se (ARGX) Stock Quote, History and News - Yahoo Finance
Argenx Se (ARGX) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...