Autolus Therapeutics plc - American Depositary Share (AUTL): Price and Financial Metrics
AUTL Price/Volume Stats
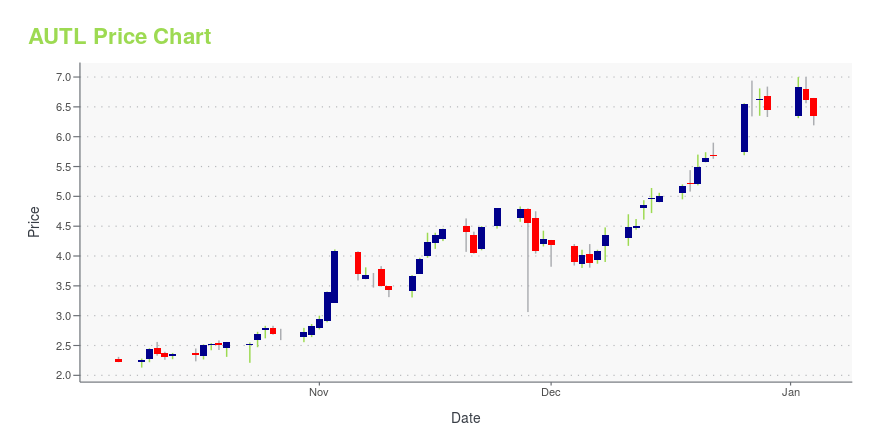
| Current price | $4.67 | 52-week high | $7.45 |
| Prev. close | $4.60 | 52-week low | $2.01 |
| Day low | $4.53 | Volume | 626,708 |
| Day high | $4.77 | Avg. volume | 1,591,024 |
| 50-day MA | $4.04 | Dividend yield | N/A |
| 200-day MA | $4.79 | Market Cap | 1.24B |
AUTL Stock Price Chart Interactive Chart >
Autolus Therapeutics plc - American Depositary Share (AUTL) Company Bio
Autolus Therapeutics is developing a pipeline of advanced programmable T cell treatment candidates for blood and solid tumor cancers. Autolus‘ T cell therapies target the the blood cancer ALL either through CD19 (in Phase I), or through targeting CD19 and CD22 (Phase I/II). The company's CD19 candidate is claimed "next-generation" due to its fast CD19 dissociation kinetics, which allows to target many CD19+ cells very rapidly. Furthermore, the company’s CAR-T cells target CD269 and TACI to treat multiple myeloma (Phase I/II). An additional candidate undergoing Phase I safety testing targets GD2, a disialoganglioside expressed on tumours of neuroectodermal origin, to treat the rare childhood brain cancer neuroblastoma. The company is based in London, England.
Latest AUTL News From Around the Web
Below are the latest news stories about AUTOLUS THERAPEUTICS PLC that investors may wish to consider to help them evaluate AUTL as an investment opportunity.
Autolus Therapeutics Announces Changes to its Board of Directors- Appointment of Elisabeth (“Lis”) Leiderman, M.D. - Resignation of Kapil Dhingra, M.D. LONDON, Dec. 22, 2023 (GLOBE NEWSWIRE) -- Autolus Therapeutics plc (Nasdaq: AUTL), a clinical-stage biopharmaceutical company developing next-generation programmed T cell therapies, today announced the appointment of Elisabeth Leiderman, M.D. to the Board of Directors. Additionally, the Company announced that Kapil Dhingra M.D., who has served on Autolus’ Board of Directors since May 2015, has advised the Boa |
12 Most Promising Cancer Stocks According to AnalystsIn this piece, we will take a look at the 12 most promising cancer stocks according to analysts. If you want to skip our overview of the latest trends in the cancer treatment sector, then you can take a look at the 5 Most Promising Cancer Stocks According to Analysts. The advent of technology and […] |
Autolus Therapeutics Presents Clinical Data Updates at the American Society of Hematology (ASH) Annual Meeting 2023Pooled analysis of the FELIX Phase Ib/II study demonstrated prolonged event free survival and low overall immunotoxicity across all cohorts in r/r B-ALL, and particularly in patients with low leukemic burden at lymphodepletionLonger-term data from a pooled analysis from the ALLCAR19 study and FELIX Phase Ib in r/r B-ALL showed durable remissions with obe-cel as a stand-alone therapy in a subset of patients after a median follow up of >3 yearsAdditionally, ALLCAR19 extension cohorts demonstrated |
Autolus Therapeutics Submits Biologics License Application to U.S. Food and Drug Administration for obecabtagene autoleucel (obe-cel) for Patients with Relapsed/refractory (r/r) Adult B-Cell Acute Lymphoblastic Leukemia (ALL)BLA submission includes results from pivotal Phase 2 FELIX study evaluating obe-cel in relapsed/refractory (r/r) adult B-cell Acute Lymphoblastic Leukemia (ALL)Company on track to submit a marketing authorization application to the European Medicines Agency (EMA) in the first half of 2024 LONDON, Nov. 27, 2023 (GLOBE NEWSWIRE) -- Autolus Therapeutics plc (Nasdaq: AUTL), a clinical-stage biopharmaceutical company developing next-generation programmed T cell therapies, today announces that it has |
Autolus Therapeutics to Host Analyst / Investor event at the American Society of Hematology (ASH) Annual Meeting 2023In-person and webcast event on Sunday, 10 December, 2023 at 8:00 AM PT / 4:00 PM GMTReview of pooled analysis of the ongoing FELIX Phase Ib/II study, pooled analysis from ALLCAR19 and FELIX Phase Ib studies and ALLCAR19 extension with obe-cel, as well as initial data from the AUTO8 MCARTY Phase I Study LONDON, Nov. 15, 2023 (GLOBE NEWSWIRE) -- Autolus Therapeutics plc (Nasdaq: AUTL), a clinical-stage biopharmaceutical company developing next-generation programmed T cell therapies, will host an i |
AUTL Price Returns
| 1-mo | 38.99% |
| 3-mo | 16.75% |
| 6-mo | -21.38% |
| 1-year | 72.96% |
| 3-year | -12.55% |
| 5-year | -66.04% |
| YTD | -27.48% |
| 2023 | 238.95% |
| 2022 | -63.39% |
| 2021 | -41.95% |
| 2020 | -32.27% |
| 2019 | -59.81% |

Loading social stream, please wait...