Biomerica, Inc. (BMRA): Price and Financial Metrics
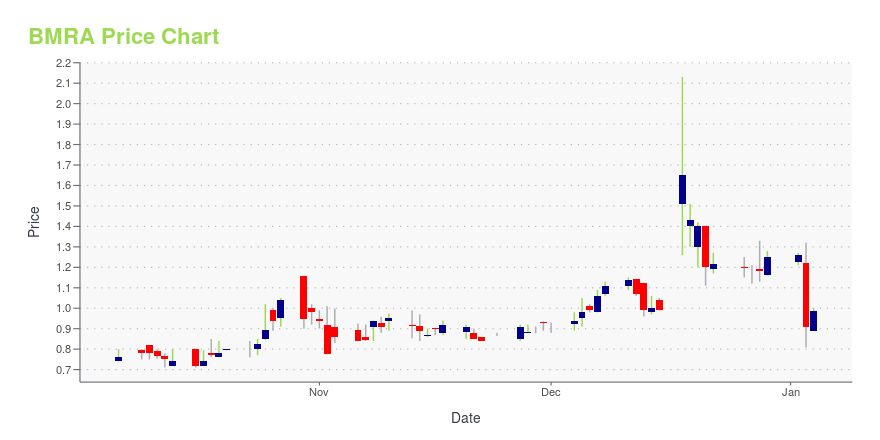
BMRA Price/Volume Stats
| Current price | $0.35 | 52-week high | $2.13 |
| Prev. close | $0.35 | 52-week low | $0.28 |
| Day low | $0.35 | Volume | 18,208 |
| Day high | $0.36 | Avg. volume | 151,647 |
| 50-day MA | $0.49 | Dividend yield | N/A |
| 200-day MA | $0.82 | Market Cap | 5.84M |
BMRA Stock Price Chart Interactive Chart >
Biomerica, Inc. (BMRA) Company Bio
Biomerica, Inc., a biomedical technology company, together with its subsidiaries, develops, patents, manufactures, and markets diagnostic and therapeutic products or detection and/or treatment of medical conditions and diseases worldwide. The company's diagnostic test kits are used to analyze blood, urine, or fecal specimens from patients in the diagnosis of various diseases and other medical complications; or to measure the level of specific bacteria, hormones, antibodies, antigens, or other substances, which exist in the patient's body and stools or blood in extremely small concentrations. It primarily sells its products for gastrointestinal diseases, food intolerances, diabetes, and various esoteric tests at the physicians' offices and over-the-counter drugstores, and hospital/clinical laboratories. The company is also developing InFoods, an irritable bowel syndrome (IBS) therapy technology and diagnostic-guided therapy that is designed to allow physicians to identify specific foods that when removed from the patient's diet can alleviate the patient's IBS symptoms; Helicobacter pylori products; and develops, tests, validates, and sells diagnostic products for COVID-19 infection. Biomerica, Inc. was founded in 1971 and is headquartered in Irvine, California.
Latest BMRA News From Around the Web
Below are the latest news stories about BIOMERICA INC that investors may wish to consider to help them evaluate BMRA as an investment opportunity.
Is Biomerica (NASDAQ:BMRA) In A Good Position To Deliver On Growth Plans?Just because a business does not make any money, does not mean that the stock will go down. For example, although... |
Today’s Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on TuesdayIt's time to dive into the biggest pre-market stock movers as we check out all of the most recent news for Tuesday morning! |
Biomerica Received US FDA 510(k) Clearance for Their Hp Detect™ ELISA Test Product Designed to Detect the Presence of the H. pylori Bacteria That infects Approximately 35% of the U.S. PopulationOver 80% of gastric cancers are attributed to H. pylori bacterial infection Gastric cancer is the third most common cause of cancer related death in the world H. pylori infection is also a major cause of peptic ulcers IRVINE, Calif., Dec. 18, 2023 (GLOBE NEWSWIRE) -- Biomerica, Inc. (Nasdaq: BMRA), (the “Company”) a global provider of advanced medical diagnostic products, today announced it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for its Hp Detect™ Stool Antigen ELI |
Biomerica to Participate in 14th Annual Craig-Hallum Alpha Select Conference on November 16IRVINE, Calif., Nov. 10, 2023 (GLOBE NEWSWIRE) -- Biomerica, Inc. (Nasdaq: BMRA), (the “Company”) a global provider of advanced medical products today announced that it will participate in the 14th Annual Craig-Hallum Alpha Select Conference on November 16, 2023, at the Sheraton New York Times Square Hotel in New York City. Zackary Irani, Chief Executive Officer of Biomerica will be hosting meetings during the event, which consists of one-on-one and small group meetings. About Biomerica (NASDAQ: |
Biomerica Expands inFoods IBS Reach with Addition of Several New GI GroupsBiomerica remains on track for inFoods® IBS national rollout during calendar 2024, and expects strong product revenue growth during national rolloutVeteran Vice President of Sales and National Accounts hired along with first set of regional sales representatives IRVINE, Calif., Nov. 08, 2023 (GLOBE NEWSWIRE) -- Biomerica, Inc. (Nasdaq: BMRA), (the “Company”) a global provider of advanced medical products today announced that several new Gastrointestinal (GI) groups have signed up and begun using |
BMRA Price Returns
| 1-mo | -22.91% |
| 3-mo | -47.38% |
| 6-mo | -61.30% |
| 1-year | -74.64% |
| 3-year | -90.14% |
| 5-year | -88.29% |
| YTD | -72.00% |
| 2023 | -62.69% |
| 2022 | -13.88% |
| 2021 | -21.89% |
| 2020 | 64.68% |
| 2019 | 77.88% |
Continue Researching BMRA
Here are a few links from around the web to help you further your research on Biomerica Inc's stock as an investment opportunity:Biomerica Inc (BMRA) Stock Price | Nasdaq
Biomerica Inc (BMRA) Stock Quote, History and News - Yahoo Finance
Biomerica Inc (BMRA) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...