Checkpoint Therapeutics, Inc. (CKPT): Price and Financial Metrics
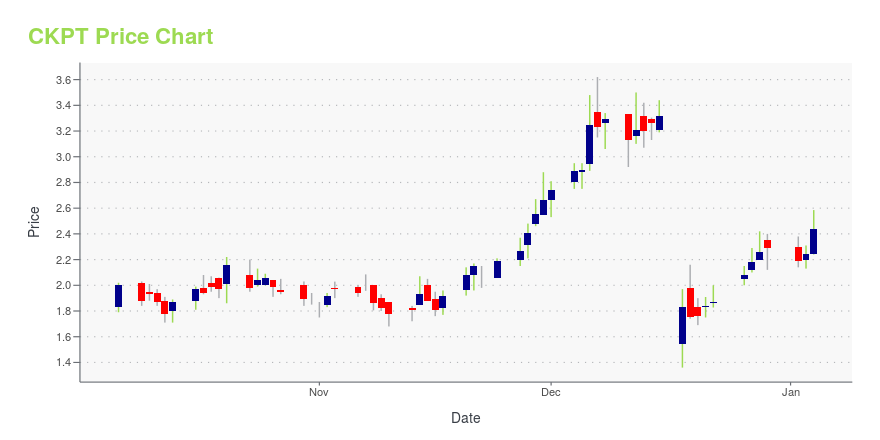
CKPT Price/Volume Stats
| Current price | $4.04 | 52-week high | $4.50 |
| Prev. close | $4.04 | 52-week low | $1.38 |
| Day low | $3.98 | Volume | 3,954,800 |
| Day high | $4.05 | Avg. volume | 2,402,256 |
| 50-day MA | $3.39 | Dividend yield | N/A |
| 200-day MA | $2.97 | Market Cap | 197.29M |
CKPT Stock Price Chart Interactive Chart >
Checkpoint Therapeutics, Inc. (CKPT) Company Bio
Checkpoint Therapeutics, Inc., an immuno-oncology biopharmaceutical company, focuses on the acquisition, development, and commercialization of non-chemotherapy and immune-enhanced combination treatments for patients with solid tumor cancers. The company was founded in 2014 and is based in New York, New York. Checkpoint Therapeutics, Inc. is a subsidiary of Fortress Biotech, Inc.
CKPT Price Returns
| 1-mo | 62.25% |
| 3-mo | 10.68% |
| 6-mo | 81.98% |
| 1-year | 116.04% |
| 3-year | -76.65% |
| 5-year | -66.33% |
| YTD | 26.25% |
| 2024 | 39.74% |
| 2023 | -55.19% |
| 2022 | -83.57% |
| 2021 | 17.36% |
| 2020 | 54.07% |
Continue Researching CKPT
Here are a few links from around the web to help you further your research on Checkpoint Therapeutics Inc's stock as an investment opportunity:Checkpoint Therapeutics Inc (CKPT) Stock Price | Nasdaq
Checkpoint Therapeutics Inc (CKPT) Stock Quote, History and News - Yahoo Finance
Checkpoint Therapeutics Inc (CKPT) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...