Centessa Pharmaceuticals Limited (CNTA): Price and Financial Metrics
CNTA Price/Volume Stats
| Current price | $10.78 | 52-week high | $12.45 |
| Prev. close | $10.29 | 52-week low | $5.15 |
| Day low | $10.18 | Volume | 822,043 |
| Day high | $10.82 | Avg. volume | 308,731 |
| 50-day MA | $9.12 | Dividend yield | N/A |
| 200-day MA | $8.72 | Market Cap | 1.08B |
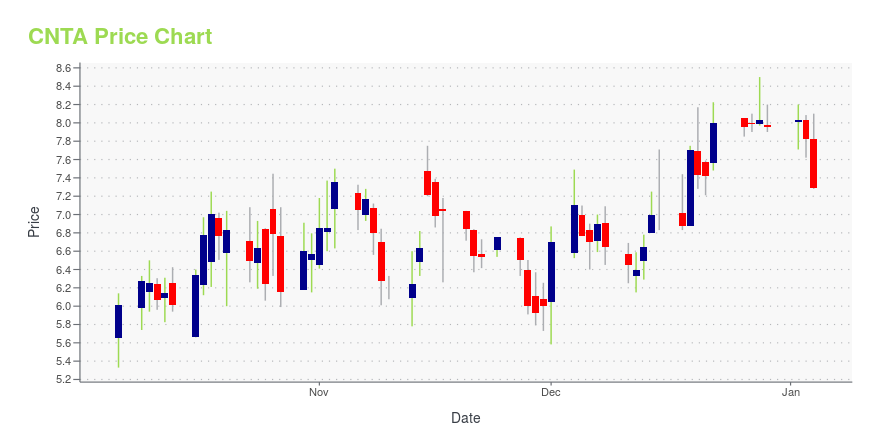
CNTA Stock Price Chart Interactive Chart >
Centessa Pharmaceuticals Limited (CNTA) Company Bio
Centessa Pharmaceuticals Limited, a pharmaceutical company, develops and delivers life-altering and life-enhancing medicines to patients. Its products pipeline include Lixivaptan, a vasopressin V2 receptor small molecule inhibitor that is in Phase 3 clinical development for the treatment of autosomal dominant polycystic kidney disease; SerpinPC, an activated protein C inhibitor, which is in Phase 2a clinical development for the treatment of hemophilia A and B; Imgatuzumab, an anti-EGFR monoclonal antibody expected to enter a Phase 2 clinical trial for the treatment of cutaneous squamous cell carcinoma, as well as is being considered for the treatment of other solid tumors in the context of combination treatment with immunotherapy; and ZF874, a small molecule chemical chaperone folding corrector of the Z variant of alpha-1-antitrypsin in Phase 1 clinical development for the treatment of alpha-1-antitrypsin deficiency. The company's preclinical product candidates comprise ZF887 for alpha-1-antitrypsin deficiency; MGX292 for pulmonary arterial hypertension; CBS001 for idiopathic pulmonary fibrosis; CBS004 for systemic sclerosis and lupus; LB1 and LB2 for solid tumors; Oral OX2R Agonist and Intranasal OX2R Agonist for narcolepsy type 1; Dual STAT3/5 Degrader for hematological malignancies; EGFR Ex20 and EGFR-C797S inhibitors for non-small cell lung cancer; and next generation EGFR inhibitors. The company was formerly known as United Medicines Biopharma Limited and changed its name to Centessa Pharmaceuticals Limited in February 2021. Centessa Pharmaceuticals Limited was incorporated in 2020 and is based in Cambridge, the United Kingdom.
Latest CNTA News From Around the Web
Below are the latest news stories about CENTESSA PHARMACEUTICALS PLC that investors may wish to consider to help them evaluate CNTA as an investment opportunity.
The Zacks Analyst Blog Highlights Amphastar Pharmaceuticals, Journey Medical, Centessa Pharmaceuticals and Aquestive TherapeuticsAmphastar Pharmaceuticals, Journey Medical, Centessa Pharmaceuticals and Aquestive Therapeutics are part of the Zacks top Analyst Blog. |
What Awaits These 4 Drug Stocks That More Than Doubled in 2023?Here are four drug, biotech stocks, AMPH, DERM, CNTA & AQST, which have more than doubled in 2023 with room for further growth in 2024. |
Centessa Pharmaceuticals to Present at the 42ⁿᵈ Annual J.P. Morgan Healthcare ConferenceBOSTON and LONDON, Dec. 13, 2023 (GLOBE NEWSWIRE) -- Centessa Pharmaceuticals plc (Nasdaq: CNTA), a clinical-stage pharmaceutical company that aims to discover and develop transformational medicines for patients, today announced its participation in the 42nd Annual J.P. Morgan Healthcare Conference in San Francisco, CA. Saurabh Saha MD PhD, Chief Executive Officer of Centessa, is scheduled to present at the conference on Wednesday, January 10, 2024, at 4:30 PM PST. The live audio webcast of this |
Centessa Pharmaceuticals Announces New Data from an Additional 52-Weeks of Continuous Treatment from Third Year (Part 5) of Ongoing Phase 2a Study of SerpinPC for the Treatment of HemophiliaPart 5 data reinforces favorable safety and tolerability profile and long-term efficacy results for SerpinPC: Median all-bleed ABR of 1.0, a 96% reduction from prospective baseline No thromboembolic events or treatment-related sustained elevations of D-dimer observed Poster presentation at American Society of Hematology (ASH) Annual meeting Registrational PRESent-2 and PRESent-3 studies of SerpinPC in hemophilia B are ongoing BOSTON and LONDON, Dec. 10, 2023 (GLOBE NEWSWIRE) -- Centessa Pharmace |
Does Centessa Pharmaceuticals PLC Sponsored ADR (CNTA) Have the Potential to Rally 40.85% as Wall Street Analysts Expect?The average of price targets set by Wall Street analysts indicates a potential upside of 40.9% in Centessa Pharmaceuticals PLC Sponsored ADR (CNTA). While the effectiveness of this highly sought-after metric is questionable, the positive trend in earnings estimate revisions might translate into an upside in the stock. |
CNTA Price Returns
| 1-mo | 19.91% |
| 3-mo | 20.85% |
| 6-mo | 32.92% |
| 1-year | 60.18% |
| 3-year | -48.84% |
| 5-year | N/A |
| YTD | 35.43% |
| 2023 | 156.77% |
| 2022 | -72.47% |
| 2021 | N/A |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...