Diffusion Pharmaceuticals Inc. (DFFN): Price and Financial Metrics
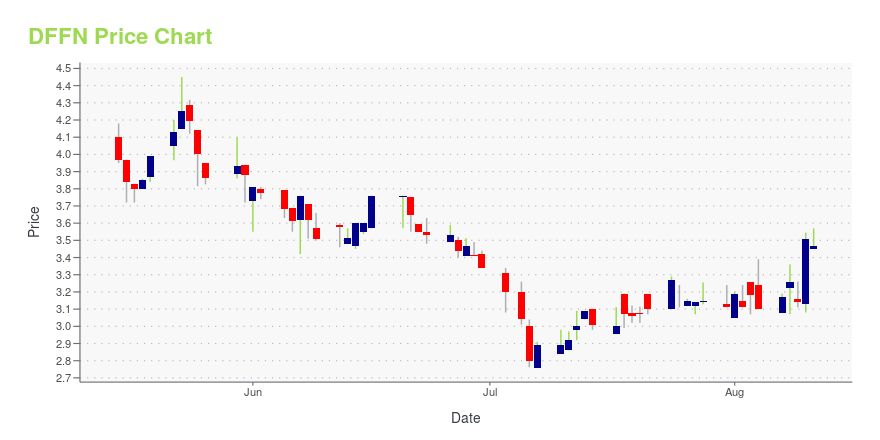
DFFN Price/Volume Stats
| Current price | $4.40 | 52-week high | $7.40 |
| Prev. close | $3.94 | 52-week low | $2.76 |
| Day low | $4.17 | Volume | 351,000 |
| Day high | $5.19 | Avg. volume | 10,937 |
| 50-day MA | $3.33 | Dividend yield | N/A |
| 200-day MA | $0.00 | Market Cap | 8.98M |
DFFN Stock Price Chart Interactive Chart >
Diffusion Pharmaceuticals Inc. (DFFN) Company Bio
Diffusion Pharmaceuticals Inc., a clinical-stage biotechnology company, focuses on developing products for ophthalmology, oncology, and dermatology. The company is based in Charlottesville, Virginia.
DFFN Price Returns
| 1-mo | N/A |
| 3-mo | N/A |
| 6-mo | N/A |
| 1-year | N/A |
| 3-year | -47.49% |
| 5-year | 266.67% |
| YTD | N/A |
| 2024 | N/A |
| 2023 | 0.00% |
| 2022 | 1,558.13% |
| 2021 | -61.64% |
| 2020 | 72.56% |

Loading social stream, please wait...