Eloxx Pharmaceuticals, Inc. (ELOX): Price and Financial Metrics
ELOX Price/Volume Stats
| Current price | $0.70 | 52-week high | $7.96 |
| Prev. close | $0.68 | 52-week low | $0.40 |
| Day low | $0.52 | Volume | 18,400 |
| Day high | $0.70 | Avg. volume | 7,950 |
| 50-day MA | $0.77 | Dividend yield | N/A |
| 200-day MA | $1.21 | Market Cap | 2.20M |
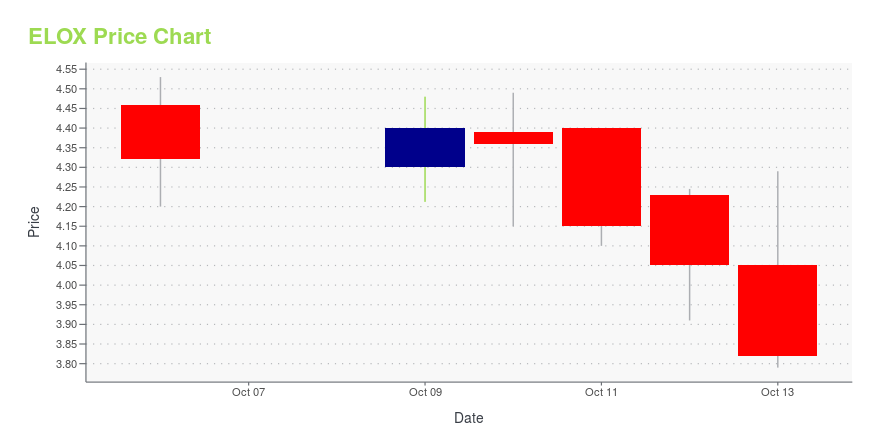
ELOX Stock Price Chart Interactive Chart >
Eloxx Pharmaceuticals, Inc. (ELOX) Company Bio
Eloxx Pharmaceuticals, Inc., a clinical-stage biopharmaceutical company, focuses on discovering and developing novel therapeutics for the treatment of rare and ultra-rare premature stop codon diseases. Its lead program is ELX-02, which is in Phase I clinical trial, which focuses on the treatment of cystic fibrosis and cystinosis patients with diagnosed nonsense mutations. The company was founded in 2013 and is based in Waltham, Massachusetts.
Latest ELOX News From Around the Web
Below are the latest news stories about ELOXX PHARMACEUTICALS INC that investors may wish to consider to help them evaluate ELOX as an investment opportunity.
Eloxx Pharmaceuticals Reports Additional Confirmation that All Nonsense Mutation Alport Syndrome Patients Treated with ELX-02 in Phase 2 Study had Improvement in Kidney Morphology and Clinical Benefit of Reduction or Stabilization of ProteinuriaPodocyte Exact Morphology Procedure Results Podocyte Exact Morphology Procedure Results ELX-02 treatment improved podocyte foot process effacement in all three patients by an average of 60% based on a blinded kidney biopsy analysis by NIPOKA GmbH Biopsy results support clinical benefit in all three patients as improvement of kidney morphology is consistent with reduction or stabilization of proteinuria during or up to 2 months post completion of dosing Renowned key opinion leaders recommend cont |
Eloxx Pharmaceuticals Announces $2 Million Registered Direct Offering Priced At-the-Market Under Nasdaq RulesWATERTOWN, Mass., Sept. 19, 2023 (GLOBE NEWSWIRE) -- Eloxx Pharmaceuticals, Inc. (NASDAQ: ELOX), a leader in ribosomal RNA-targeted genetic therapies for rare diseases, today announced that it has entered into a definitive agreement for the issuance and sale of an aggregate of 380,590 of its shares of common stock (or common stock equivalents in lieu thereof) at a purchase price of $5.255 per share (or common stock equivalent in lieu thereof) in a registered direct offering priced at-the-market |
Eloxx Pharmaceuticals Reports Independent Confirmation of Positive Biopsy Results in All Patients Treated with ELX-02 in Phase 2 Clinical Study for Alport SyndromeHighly regarded renal pathologist and transmission electron microscopy (TEM) expert independently confirms previously reported qualitative assessment by Mayo Clinic of TEM biopsy scans All three patients treated with ELX-02 showed a visual improvement in podocyte foot process effacement post-treatment in kidney biopsies demonstrating the disease modifying effect of ELX-02 Podocyte foot process effacement is a hallmark of Alport syndrome Eloxx intends to gain alignment with U.S. Food and Drug Adm |
Eloxx Pharmaceuticals Provides Program Updates on ELX-02 and ZKN-013Investigational New Drug (IND) application submitted to U.S. Food and Drug Administration (FDA) for ELX-02 for the treatment of Alport syndrome with nonsense mutations Rebound in average UPCR 3-months post treatment provides further evidence that proteinuria remission in one out of three patients was drug related All 3 Alport patients (100% response rate) treated with ELX-02 had biopsy confirmed disease regression, suggesting clinical benefit likely with longer treatment duration Significant str |
Eloxx Pharmaceuticals Reports Drug Response in All Patients Treated with ELX-02 in Phase 2 Clinical Study for Alport SyndromeAll three patients (100% response rate) treated with ELX-02 showed an improvement in podocyte foot process effacement post-treatment in kidney biopsies assessed by electron microscopy demonstrating the disease modifying effect of ELX-02 Podocyte foot process effacement is a hallmark of Alport syndrome Together with previously announced rapid and sustained proteinuria remission in one patient in Phase 2 clinical study, these biopsy results strongly support decision to advance ELX-02 into a pivota |
ELOX Price Returns
| 1-mo | 1.45% |
| 3-mo | -13.13% |
| 6-mo | -16.17% |
| 1-year | -88.29% |
| 3-year | -98.73% |
| 5-year | -99.78% |
| YTD | -41.67% |
| 2023 | -34.07% |
| 2022 | -93.56% |
| 2021 | -82.26% |
| 2020 | -45.92% |
| 2019 | -38.72% |

Loading social stream, please wait...