Eton Pharmaceuticals, Inc. (ETON): Price and Financial Metrics
ETON Price/Volume Stats
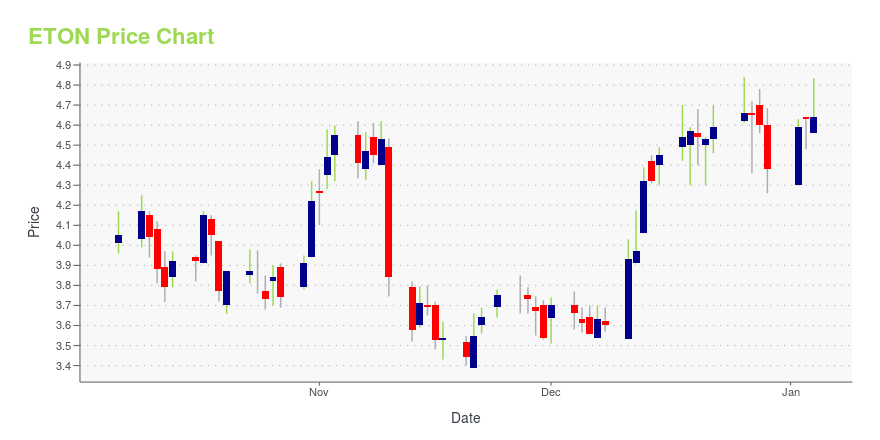
| Current price | $3.58 | 52-week high | $5.81 |
| Prev. close | $3.57 | 52-week low | $2.42 |
| Day low | $3.58 | Volume | 18,513 |
| Day high | $3.65 | Avg. volume | 59,302 |
| 50-day MA | $3.50 | Dividend yield | N/A |
| 200-day MA | $3.90 | Market Cap | 91.97M |
ETON Stock Price Chart Interactive Chart >
Eton Pharmaceuticals, Inc. (ETON) Company Bio
Eton Pharmaceuticals Inc. Eton Pharmaceuticals, Inc. is a is a specialty pharmaceutical company, which is focused on developing and commercializing pharmaceutical products. It products include EM-100, ET-103, CT-100, DS-300, DS-200, and DS-100. The Company focuses on product candidates that are liquid in formulation, including injections, oral liquids and ophthalmics. EM-100 is an ophthalmic product with a preservative-free formulation of the active ingredient ketotifen, and is indicated for the treatment of allergic conjunctivitis. ET-103 is an oral liquid formulation of levothyroxine, which is widely used in tablet and capsule form, and is indicated for the treatment of hypothyroidism. CT-100 is our patent-pending synthetic corticotropin therapeutic candidate that mimics the amino acid chain of injectable product H.P. Acthar Gel. DS-300 is a patent-pending injectable product candidate indicated to treat nutritional deficiencies. DS-200 is an injectable product candidate indicated to treat pain management.
Latest ETON News From Around the Web
Below are the latest news stories about ETON PHARMACEUTICALS INC that investors may wish to consider to help them evaluate ETON as an investment opportunity.
5 Under-$15 Stocks Wall Street Analysts Recommend BuyingAfter a stellar rally in Wall Street this year, investors with limited funds but desire to participate in the market can bet on low-priced top-ranked stocks like Eton Pharmaceuticals (ETON), LegalZoom.com (LZ), PLAYSTUDIOS (MYPS), American Public Education (APEI) and Cellebrite DI (CLBT). |
One Eton Pharmaceuticals, Inc. (NASDAQ:ETON) Analyst Just Made A Major Cut To Next Year's EstimatesMarket forces rained on the parade of Eton Pharmaceuticals, Inc. ( NASDAQ:ETON ) shareholders today, when the covering... |
Eton Pharmaceuticals, Inc. (NASDAQ:ETON) Q3 2023 Earnings Call TranscriptEton Pharmaceuticals, Inc. (NASDAQ:ETON) Q3 2023 Earnings Call Transcript November 9, 2023 Eton Pharmaceuticals, Inc. beats earnings expectations. Reported EPS is $-0.02, expectations were $-0.1. Operator: Good afternoon, and welcome to the Eton Pharmaceuticals Third Quarter 2023 Financial Results Conference Call. [Operator Instructions] Please be advised that this call is being recorded at the company’s […] |
Declining Stock and Solid Fundamentals: Is The Market Wrong About Eton Pharmaceuticals, Inc. (NASDAQ:ETON)?Eton Pharmaceuticals (NASDAQ:ETON) has had a rough week with its share price down 16%. But if you pay close attention... |
Eton Pharmaceuticals Reports Third Quarter 2023 Financial ResultsProduct sales and royalty revenue of $7.0 million up 118% from Q3 2022, representing the eleventh straight quarter of sequential product sales and royalty revenue growthGenerated $0.9 million in positive cash flow from operations; ended quarter with $22.1 million of cash on handAcquired ultra-rare disease product Nitisinone CapsulesManagement to hold conference call today at 4:30pm ET DEER PARK, Ill., Nov. 09, 2023 (GLOBE NEWSWIRE) -- Eton Pharmaceuticals, Inc (“Eton” or “the Company”) (Nasdaq: |
ETON Price Returns
| 1-mo | 6.55% |
| 3-mo | 3.77% |
| 6-mo | -21.49% |
| 1-year | 31.62% |
| 3-year | -33.58% |
| 5-year | -42.81% |
| YTD | -18.26% |
| 2023 | 55.32% |
| 2022 | -34.27% |
| 2021 | -47.23% |
| 2020 | 12.92% |
| 2019 | 17.65% |
Continue Researching ETON
Want to do more research on Eton Pharmaceuticals Inc's stock and its price? Try the links below:Eton Pharmaceuticals Inc (ETON) Stock Price | Nasdaq
Eton Pharmaceuticals Inc (ETON) Stock Quote, History and News - Yahoo Finance
Eton Pharmaceuticals Inc (ETON) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...