GENFIT S.A. (GNFT): Price and Financial Metrics
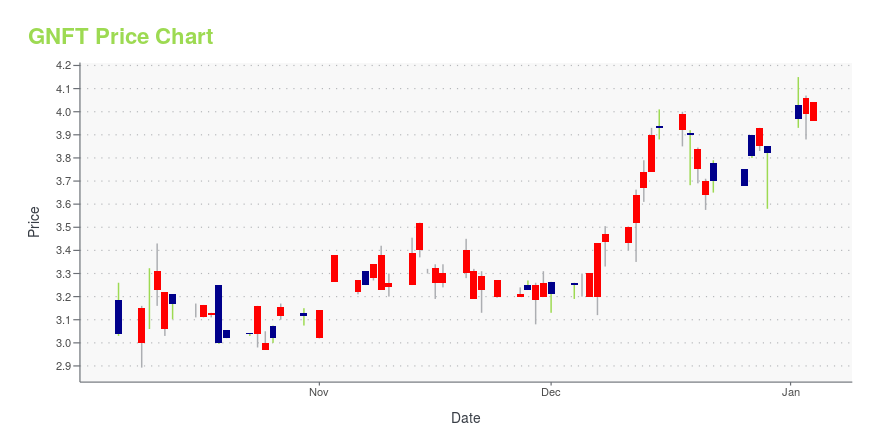
GNFT Price/Volume Stats
| Current price | $4.01 | 52-week high | $6.05 |
| Prev. close | $4.52 | 52-week low | $2.89 |
| Day low | $3.84 | Volume | 20,216 |
| Day high | $4.40 | Avg. volume | 18,779 |
| 50-day MA | $4.43 | Dividend yield | N/A |
| 200-day MA | $3.75 | Market Cap | 200.12M |
GNFT Stock Price Chart Interactive Chart >
GENFIT S.A. (GNFT) Company Bio
Genfit SA is a biopharmaceutical company involved in drug discovery and development for the early diagnosis, prevention and treatment of cardiometabolic diseases. The company focuses on the discovery and development of drug candidates in areas of high unmet medical needs corresponding to a lack of suitable treatments. It focuses on medicines to market for patients with metabolic, inflammatory, autoimmune and fibrotic diseases, that affect the liver. The company was founded by Jean-François Mouney, Florence Séjourné and Bart Staels on September 1999 and is headquartered in Loos, France.
Latest GNFT News From Around the Web
Below are the latest news stories about GENFIT SA that investors may wish to consider to help them evaluate GNFT as an investment opportunity.
GENFIT Updates 2024 Outlook Following Acceptance of Elafibranor Filings in Primary Biliary Cholangitis (PBC)US Food and Drug Administration (FDA) has granted Priority Review for New Drug Application (NDA) for elafibranor in PBC, and European Medicine Agency (EMA) has also validated the Marketing Authorization Application (MAA) for elafibranor.Acceptance triggers a first milestone payment. Further milestones are expected upon US and European launches which could now happen in 2Q24 in the US (FDA PDUFA1 action date: June 10, 2024) and 2H24 in Europe. These milestones total approximately 89M€. Launches i |
Ipsen confirms U.S. FDA grants priority review for New Drug Application for elafibranor for the treatment of rare cholestatic liver disease, PBCNew Drug Application granted priority review with PDUFA date set for June 10, 2024European Medicines Agency (EMA) has also validated the Marketing Authorization Application (MAA) for elafibranor Investigational elafibranor is the first novel second-line treatment for primary biliary cholangitis (PBC) to be filed in E.U. and U.S. in nearly a decade Paris (France), December 07, 2023 – Ipsen (Euronext: IPN; ADR: IPSEY) and GENFIT (Nasdaq and Euronext: GNFT) today announced that the U.S. Food and Dr |
GENFIT Announces Publication in the Journal of Hepatology on the Accurate Performance of NIS2+™ as a Screening Tool for the Enrollment of Patients in MASH Clinical TrialsThe paper published in Journal of Hepatology demonstrates that NIS2+™ technology could significantly reduce liver biopsy failure rate and maximizes accuracy of patient selection for MASH1 clinical trialsGrowing body of clinical evidence: this is the third paper on NIS2+™ that has been published in a leading medical journal in 2023 Lille (France), Cambridge (Massachusetts, United States), Zurich (Switzerland), December 6, 2023- GENFIT (Nasdaq and Euronext: GNFT), a late-stage biopharmaceutical co |
GENFIT Highlights ACLF Development Strategy at “ACLF Day” during AASLD The Liver Meeting® 2023GENFIT highlighted new focus and development strategy in Acute On-Chronic Liver Failure (ACLF)ACLF is a very serious condition affecting ~294,0001 2 patients with chronic liver diseases in the USA and Europe every year and is associated with multi-organ failure and high short-term mortalityIt is an underserved medical condition with currently no approved treatment and few programs under development globallyGENFIT has developed a unique pipeline of 5 different drug candidates targeting key pathop |
GENFIT: Results from Ipsen’s ELATIVE® pivotal Phase III trial of elafibranor in PBC presented as late breaking data at AASLD congress and published in New England Journal of MedicineELATIVE® Phase III trial confirms potential for investigational elafibranor as a novel, first-in-class, dual PPAR α,δ agonist for patients with Primary Biliary Cholangitis.Elafibranor demonstrates significant improvements in biomarkers of disease progression versus placebo, including significant treatment benefit with improvement in biochemical response and alkaline phosphatase (ALP) normalization, along with patient-reported outcomes data suggesting a possible improvement in pruritus.Elafibrano |
GNFT Price Returns
| 1-mo | 6.93% |
| 3-mo | 14.90% |
| 6-mo | 4.70% |
| 1-year | 8.97% |
| 3-year | 8.97% |
| 5-year | -77.19% |
| YTD | 4.16% |
| 2023 | -12.28% |
| 2022 | -12.22% |
| 2021 | 4.17% |
| 2020 | -75.88% |
| 2019 | N/A |

Loading social stream, please wait...