IO Biotech, Inc. (IOBT): Price and Financial Metrics
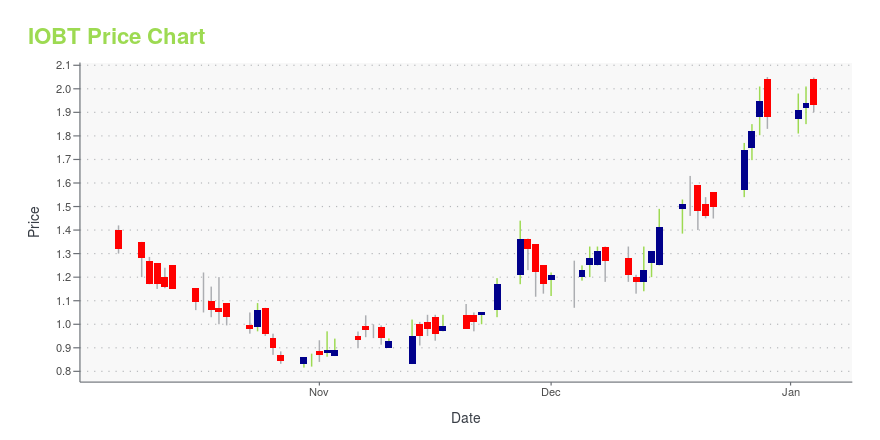
IOBT Price/Volume Stats
| Current price | $1.29 | 52-week high | $2.18 |
| Prev. close | $1.34 | 52-week low | $0.82 |
| Day low | $1.28 | Volume | 116,233 |
| Day high | $1.36 | Avg. volume | 132,700 |
| 50-day MA | $1.28 | Dividend yield | N/A |
| 200-day MA | $1.43 | Market Cap | 84.99M |
IOBT Stock Price Chart Interactive Chart >
IO Biotech, Inc. (IOBT) Company Bio
IO Biotech, Inc., a clinical-stage biopharmaceutical company, develops disruptive immune modulating anti-cancer therapies in Denmark. The company's lead product candidate, IO102-IO103, is designed to target immunosuppressive proteins, such as Indoleamine 2,3-dehydrogenase (IDO), and programmed death ligand (PD-L1). It also develops IO112, a product candidate that contains a single Arginase 1-derived peptide designed to target T cells that recognize epitopes derived from Arginase 1 for the treatment of cancers. IO Biotech, Inc. was incorporated in 2021 and is based in Copenhagen, Denmark.
Latest IOBT News From Around the Web
Below are the latest news stories about IO BIOTECH INC that investors may wish to consider to help them evaluate IOBT as an investment opportunity.
IO Biotech Announces First Patient Dosed in Phase 2 Neoadjuvant/Adjuvant Solid Tumor Basket TrialNEW YORK, Dec. 21, 2023 (GLOBE NEWSWIRE) -- IO Biotech (Nasdaq: IOBT), a clinical-stage biopharmaceutical company developing novel, immune modulating therapeutic cancer vaccines based on its T-win® platform, announced today that the first patient has been dosed in its Phase 2 trial (NCT05280314) studying treatment with IO102-IO103 in combination with Merck’s anti-PD-1 therapy KEYTRUDA® (pembrolizumab) in neoadjuvant and adjuvant patients with resectable melanoma or squamous cell carcinoma of the |
We're Keeping An Eye On IO Biotech's (NASDAQ:IOBT) Cash Burn RateWe can readily understand why investors are attracted to unprofitable companies. For example, although... |
IO Biotech Announces 2023 Third Quarter ResultsCompleted enrollment of 380 patients in pivotal Phase 3 trial of lead therapeutic cancer vaccine, IO102-IO103 in combination with KEYTRUDA® (pembrolizumab), in patients with advanced melanoma Presented encouraging preliminary data from Phase 2 basket trial of IO102-IO103 at the IASLC 2023 World Conference on Lung Cancer (WCLC) in September 2023 and at the European Society of Medical Oncology (ESMO) annual meeting in October 2023Presented three posters at the Society for Immunotherapy of Cancer’s |
IO Biotech Appoints Helen Collins, MD, to its Board of DirectorsNEW YORK, Nov. 13, 2023 (GLOBE NEWSWIRE) -- IO Biotech (Nasdaq: IOBT), a clinical-stage biopharmaceutical company developing novel, immune modulating cancer vaccines based on its T-win® technology platform, announced the appointment of Helen Collins, MD, to its board of directors. Dr. Collins is an industry veteran with significant experience in oncology/hematology drug development and will serve on the Research and Development Committee of the Board. “We are pleased to welcome Helen, an accompl |
IO Biotech Completes Enrollment of 380 Patients in Pivotal Phase 3 Trial in Advanced MelanomaTrial expected to reach the primary endpoint of progression free survival in the second half of 2025 Per-protocol interim analysis expected in mid-2024 NEW YORK, Nov. 10, 2023 (GLOBE NEWSWIRE) -- IO Biotech (Nasdaq: IOBT), a clinical-stage biopharmaceutical company developing novel, immune-modulating therapeutic cancer vaccines, announced today that it has completed enrollment in the pivotal Phase 3 clinical trial for IO102-IO103, in combination with KEYTRUDA® (pembrolizumab), Merck’s anti-PD-1 |
IOBT Price Returns
| 1-mo | 9.32% |
| 3-mo | -9.15% |
| 6-mo | -27.93% |
| 1-year | -32.81% |
| 3-year | N/A |
| 5-year | N/A |
| YTD | -31.38% |
| 2023 | -18.26% |
| 2022 | -64.06% |
| 2021 | N/A |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...