KemPharm, Inc. (KMPH): Price and Financial Metrics
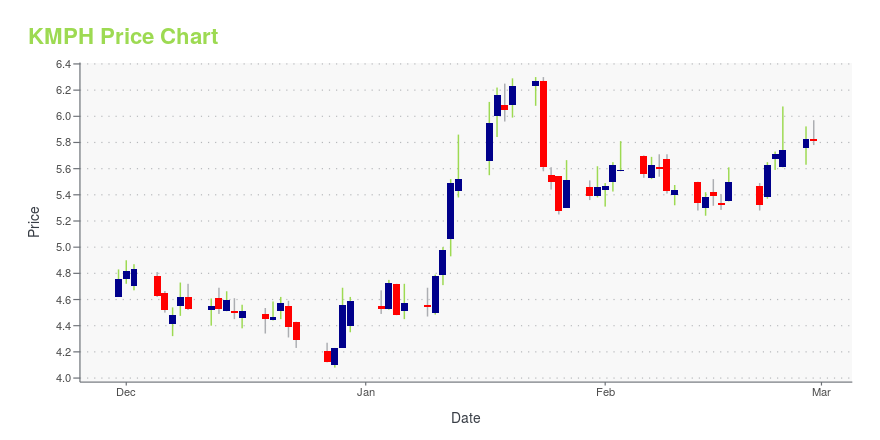
KMPH Price/Volume Stats
| Current price | $5.81 | 52-week high | $6.92 |
| Prev. close | $5.83 | 52-week low | $4.00 |
| Day low | $5.78 | Volume | 64,100 |
| Day high | $5.97 | Avg. volume | 189,500 |
| 50-day MA | $5.22 | Dividend yield | N/A |
| 200-day MA | $5.25 | Market Cap | 200.47M |
KMPH Stock Price Chart Interactive Chart >
KemPharm, Inc. (KMPH) Company Bio
KemPharm Inc. provides biopharmaceutical services. The Company offers discovery and development of therapies for the treatment of attention deficit hyperactivity disorder (ADHD), pain, and cardiovascular diseases.
Latest KMPH News From Around the Web
Below are the latest news stories about KEMPHARM INC that investors may wish to consider to help them evaluate KMPH as an investment opportunity.
Zevra Therapeutics Begins Trading as ZVRACompany’s common stock will begin trading on the Nasdaq Global Select Market under the new ticker symbol “ZVRA” starting today, March 1, 2023 New Company name, Zevra, is Greek for zebra, the internationally recognized symbol for the rare disease community CELEBRATION, Fla., March 01, 2023 (GLOBE NEWSWIRE) -- Zevra Therapeutics (NasdaqGS: ZVRA) (“Zevra” or the “Company” and formerly KemPharm, Inc.), a rare disease therapeutics company, announced today that it will begin trading under the new tick |
Zevra Therapeutics Reiterates Commitment to Rare Disease Community as New Corporate Council Member of the National Organization for Rare Disorders (NORD)CELEBRATION, Fla., Feb. 28, 2023 (GLOBE NEWSWIRE) -- Zevra Therapeutics, Inc. (NasdaqGS: KMPH) (“Zevra” or the “Company” and formerly KemPharm, Inc.), a rare disease therapeutics company, today announced that it has become a Corporate Council member of the National Organization for Rare Disorders (NORD) and reiterated its commitment to the rare disease community by celebrating Rare Disease Day 2023. A rare disease is a condition that impacts fewer than 200,000 people in the United States, with o |
Zevra Therapeutics, a Rare Disease Therapeutics Company, Files Preliminary ProxyCELEBRATION, Fla., Feb. 27, 2023 (GLOBE NEWSWIRE) -- Zevra Therapeutics, Inc. (NasdaqGS: KMPH) (“Zevra” or the “Company” and formerly known as KemPharm, Inc.), today announced it has filed its preliminary proxy statement with the U.S. Securities and Exchange Commission (the “SEC”) in connection with the Company’s 2023 Annual Meeting of Stockholders (“Annual Meeting”), which is scheduled to take place on April 25, 2023. Stockholders do not need to take any action with respect to the 2023 Annual M |
Zevra Therapeutics to Report Fourth Quarter and Full-Year 2022 ResultsCELEBRATION, Fla., Feb. 27, 2023 (GLOBE NEWSWIRE) -- Zevra Therapeutics, Inc. (NasdaqGS: KMPH) (Zevra, or the Company, formerly KemPharm, Inc.), a rare disease therapeutics company, has announced that the Company will host a conference call and live audio webcast on Tuesday, March 7, 2023, at 8:30 a.m. ET, to discuss its corporate and financial results for the fourth quarter and full-year 2022. The audio webcast with a slide presentation will be accessible via the Investor Relations section of t |
Zevra Therapeutics Announces Arimoclomol Research Featured in Two Poster Presentations at the 19th Annual WORLDSymposium™ 2023Interim data analysis of four-year, open-label extension from Phase 2/3 clinical trial suggests arimoclomol may reduce long-term progression of Niemann-Pick Disease Type C (NPC) CELEBRATION, Fla., Feb. 24, 2023 (GLOBE NEWSWIRE) -- Zevra Therapeutics, Inc. (NasdaqGS: KMPH) (“Zevra” or the “Company,” formerly known as KemPharm, Inc.), today announced that arimoclomol, the Company’s orally-delivered, first-in-class investigational product candidate being developed as a treatment for Niemann-Pick di |
KMPH Price Returns
| 1-mo | N/A |
| 3-mo | N/A |
| 6-mo | N/A |
| 1-year | N/A |
| 3-year | -46.00% |
| 5-year | -73.69% |
| YTD | N/A |
| 2023 | 0.00% |
| 2022 | -47.30% |
| 2021 | -22.23% |
| 2020 | 84.55% |
| 2019 | -78.69% |

Loading social stream, please wait...