Karuna Therapeutics, Inc. (KRTX): Price and Financial Metrics
KRTX Price/Volume Stats
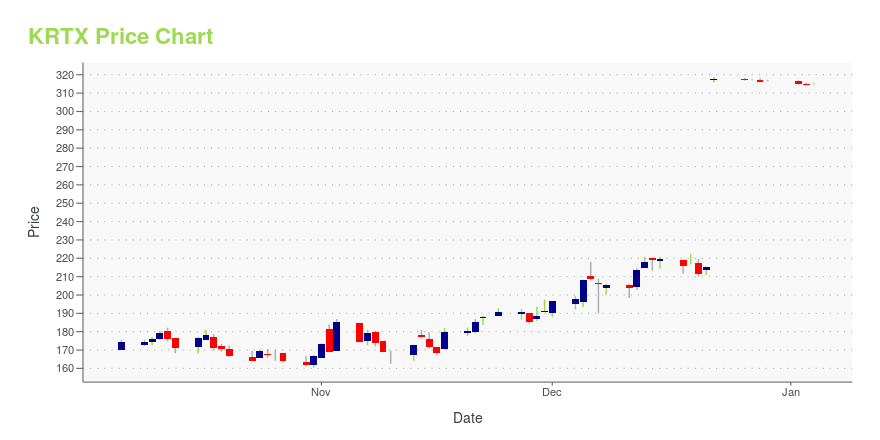
| Current price | $329.83 | 52-week high | $329.99 |
| Prev. close | $329.74 | 52-week low | $158.38 |
| Day low | $329.75 | Volume | 1,868,500 |
| Day high | $329.99 | Avg. volume | 832,048 |
| 50-day MA | $317.38 | Dividend yield | N/A |
| 200-day MA | $228.31 | Market Cap | 12.58B |
KRTX Stock Price Chart Interactive Chart >
Karuna Therapeutics, Inc. (KRTX) Company Bio
Karuna Therapeutics, Inc. operates as a clinical-stage biopharmaceutical company. It engages in business of research and development of therapies utilizing muscarinic cholinergic receptors to treat psychosis and cognitive impairment in numerous central nervous system disorders. The company develops treatments for people afflicted with schizophrenia. Karuna Therapeutics was founded in July 2009 by Andrew Miller, Eric Elenko and Peter Jeffrey Conn and is headquartered in Boston, MA.
Latest KRTX News From Around the Web
Below are the latest news stories about KARUNA THERAPEUTICS INC that investors may wish to consider to help them evaluate KRTX as an investment opportunity.
Biotech Stock Roundup: BMY's KRTX and RYZB Acquisition, CYTK Surges on Study DataBristol Myers (BMY) and Cytokinetics (CYTK) are in the news on acquisition announcements and study updates, respectively. |
Bristol Myers (BMY) to Buy Cancer Drug-Maker RayzeBio for $4.1BBristol Myers (BMY) is set to acquire RayzeBio for $4.1 billion to strengthen and diversify its already established oncology portfolio. The transaction is expected to close in the first half of 2024. |
Trending tickers: Intel | Tencent | RayzeBio | AstrazenecaThe latest investor updates on stocks that are trending on Wednesday. |
Why Is RayzeBio (RYZB) Stock Up 100% Today?RYZB stock has doubled today after Bristol Myers Squibb shelled out $4.1 billion to buy RayzeBio. |
Bristol Myers buying RayzeBio, FedEx buyback: Trending tickersFedEx (FDX) shares rose after the shipping giant announced a $1 billion accelerated share buyback plan. Shares of RayzeBio (RYZB) jumped 100% after it was announced the company was being acquired by Bristol Myers Squibb (BMY) in a $4.1 billion deal. It's Bristol Myers' second deal in less than a week. On Friday, the company announced it was acquiring Karuna Therapeutics (KRTX) in a $14 billion deal. For more expert insight and the latest market action, click here to watch this full episode of Yahoo Finance Live. |
KRTX Price Returns
| 1-mo | N/A |
| 3-mo | N/A |
| 6-mo | 5.04% |
| 1-year | 66.21% |
| 3-year | 196.93% |
| 5-year | 1,577.67% |
| YTD | 4.21% |
| 2023 | 61.07% |
| 2022 | 50.00% |
| 2021 | 28.95% |
| 2020 | 34.84% |
| 2019 | N/A |

Loading social stream, please wait...