Osmotica Pharmaceuticals plc - Ordinary Shares (OSMT): Price and Financial Metrics
OSMT Price/Volume Stats
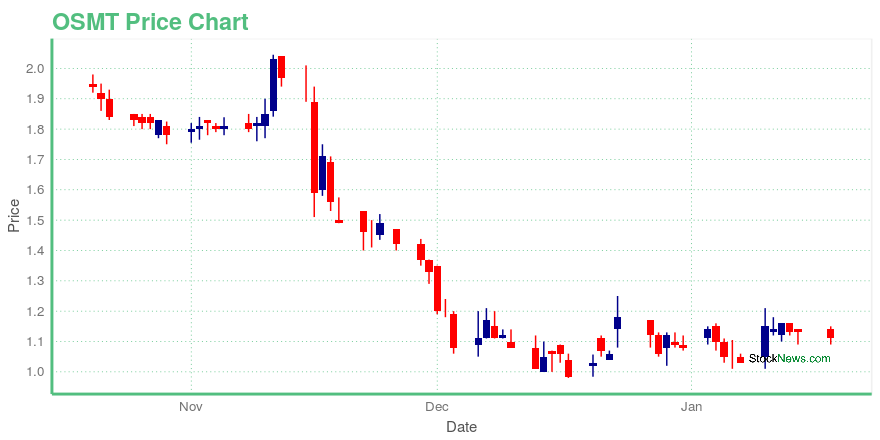
| Current price | $1.11 | 52-week high | $4.95 |
| Prev. close | $1.13 | 52-week low | $0.98 |
| Day low | $1.09 | Volume | 296,500 |
| Day high | $1.15 | Avg. volume | 396,682 |
| 50-day MA | $1.29 | Dividend yield | N/A |
| 200-day MA | $2.59 | Market Cap | 69.47M |
OSMT Stock Price Chart Interactive Chart >
Osmotica Pharmaceuticals plc - Ordinary Shares (OSMT) Company Bio
Osmotica Pharmaceuticals Plc is a holding company engages in the development and commercialization of specialty products that target markets with underserved patient populations. It offers the M-72, Lorzone which is the chlorzoxazone scored tablets and ConZip which is tramadol hydrochloride extended-release capsules in specialty neurology; and OB Complete, which is the family of prescription prenatal dietary supplements, and Divigel in women's health. The company was founded on July 13, 2017 and is headquartered in Bridgewater, NJ.
Latest OSMT News From Around the Web
Below are the latest news stories about Osmotica Pharmaceuticals plc that investors may wish to consider to help them evaluate OSMT as an investment opportunity.
RVL Pharmaceuticals to Host UPNEEQ® Overview and Business Update Webcast on January 19, 2022-- Topics include corporate name change, launch plans into the medical aesthetics market and general business updates -- -- Featured speakers include Dr. Jackie Yee and Dr. Justin Harper -- Bridgewater, N.J., Jan. 18, 2022 (GLOBE NEWSWIRE) -- Osmotica Pharmaceuticals plc (NASDAQ: OSMT) (“Osmotica” or the “Company”), a specialty pharmaceutical company, announced today that Brian Markison, Chief Executive Officer and James “JD” Schaub, Chief Operating Officer, will host a video webcast to discuss |
Osmotica Pharmaceuticals plc to Host UPNEEQ® Overview and Business Update Call on January 19, 2022BRIDGEWATER, N.J., Jan. 06, 2022 (GLOBE NEWSWIRE) -- Osmotica Pharmaceuticals plc (Nasdaq: OSMT) (“Osmotica” or the “Company”), a specialty pharmaceutical company, announced today that Brian Markison, Chief Executive Officer, James “JD” Schaub, Chief Operating Officer, and other key speakers, will host a call to discuss recent UPNEEQ® trends, pending expansion into the medical Aesthetics market and other general business updates as follows: Date:Wednesday, January 19, 2022Time:8:30 a.m. ETWebcas |
H.C. Wainwright Thinks Osmotica Pharmaceuticals’ Stock is Going to RecoverH.C. Wainwright analyst Douglas Tsao initiated coverage with a Buy rating on Osmotica Pharmaceuticals (OSMT – Research Report) today and set a price target of $5.00. The company's shares closed last Wednesday at $1.06, close to its 52-week low of $0.98. According to TipRanks.com, Tsao is a 4-star analyst with an average return of 8.8% and a 47.6% success rate. Tsao covers the Healthcare sector, focusing on stocks such as Biohaven Pharmaceutical Holding Co, Cerevel Therapeutics Holdings, and Praxis Precision Medicines. The word on The Street in general, suggests a Strong Buy analyst consensus rating for Osmotica Pharmaceuticals with a $5.75 average price target. |
Today’s Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on ThursdayGood morning, trader! |
Executive VP & COO James Schaub Just Bought 30% More Shares In Osmotica Pharmaceuticals plc (NASDAQ:OSMT)Potential Osmotica Pharmaceuticals plc ( NASDAQ:OSMT ) shareholders may wish to note that the Executive VP & COO, James... |
OSMT Price Returns
| 1-mo | N/A |
| 3-mo | N/A |
| 6-mo | N/A |
| 1-year | N/A |
| 3-year | -68.10% |
| 5-year | -66.36% |
| YTD | N/A |
| 2023 | N/A |
| 2022 | 0.00% |
| 2021 | -73.79% |
| 2020 | -41.06% |
| 2019 | -9.81% |
Continue Researching OSMT
Want to see what other sources are saying about Osmotica Pharmaceuticals plc's financials and stock price? Try the links below:Osmotica Pharmaceuticals plc (OSMT) Stock Price | Nasdaq
Osmotica Pharmaceuticals plc (OSMT) Stock Quote, History and News - Yahoo Finance
Osmotica Pharmaceuticals plc (OSMT) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...