Regeneron Pharmaceuticals Inc. (REGN): Price and Financial Metrics
REGN Price/Volume Stats
| Current price | $1,078.63 | 52-week high | $1,106.16 |
| Prev. close | $1,064.19 | 52-week low | $723.56 |
| Day low | $1,067.54 | Volume | 380,897 |
| Day high | $1,091.31 | Avg. volume | 467,901 |
| 50-day MA | $1,031.45 | Dividend yield | N/A |
| 200-day MA | $931.28 | Market Cap | 118.85B |
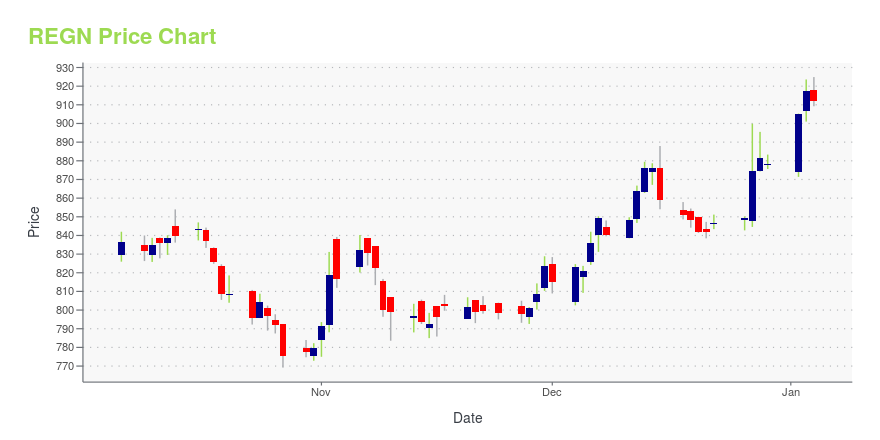
REGN Stock Price Chart Interactive Chart >
Regeneron Pharmaceuticals Inc. (REGN) Company Bio
Regeneron Pharmaceuticals, Inc. is an American biotechnology company headquartered in Westchester County, New York. The company was founded in 1988. Originally focused on neurotrophic factors and their regenerative capabilities, giving rise to its name, the company then branched out into the study of both cytokine and tyrosine kinase receptors. (Wikipedia:Source)
Latest REGN News From Around the Web
Below are the latest news stories about REGENERON PHARMACEUTICALS INC that investors may wish to consider to help them evaluate REGN as an investment opportunity.
Regeneron Gains After Eye-Drug Patent Victory Over Viatris(Bloomberg) -- Regeneron Pharmaceuticals Inc. rose the most in almost two months after a court ruled that the drugmaker’s patent rights were infringed by a cheaper copy of its eye drug developed by Viatris Inc. Most Read from BloombergThe Late-Night Email to Tim Cook That Set the Apple Watch Saga in MotionChinese Carmaker Overtakes Tesla as World’s Most Popular EV MakerBridgewater CEO’s Past Office Romance Led to Favoritism ClaimsXiaomi Unveils Its First EV, With Ambition to Be China’s Porsche o |
7 Undervalued Nasdaq-100 Stocks for Bargain HuntersUnderstandably, with the innovation sector printing a remarkable performance, all eyes have centered on Nasdaq-100 stocks. |
Where Will Regeneron Pharmaceuticals Be in 1 Year?Can the stock continue its market-beating ways? |
These 4 Measures Indicate That Regeneron Pharmaceuticals (NASDAQ:REGN) Is Using Debt Reasonably WellDavid Iben put it well when he said, 'Volatility is not a risk we care about. What we care about is avoiding the... |
The Zacks Analyst Blog Highlights Novo Nordisk, Eli Lilly, Vertex Pharmaceuticals, Regeneron and GSKNovo Nordisk, Eli Lilly, Vertex Pharmaceuticals, Regeneron and GSK are included in this Analyst Blog. |
REGN Price Returns
| 1-mo | 0.89% |
| 3-mo | 22.13% |
| 6-mo | 14.74% |
| 1-year | 44.85% |
| 3-year | 85.65% |
| 5-year | 248.87% |
| YTD | 22.81% |
| 2023 | 21.73% |
| 2022 | 14.25% |
| 2021 | 30.72% |
| 2020 | 28.66% |
| 2019 | 0.53% |
Continue Researching REGN
Want to see what other sources are saying about Regeneron Pharmaceuticals Inc's financials and stock price? Try the links below:Regeneron Pharmaceuticals Inc (REGN) Stock Price | Nasdaq
Regeneron Pharmaceuticals Inc (REGN) Stock Quote, History and News - Yahoo Finance
Regeneron Pharmaceuticals Inc (REGN) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...