Spero Therapeutics, Inc. (SPRO): Price and Financial Metrics
SPRO Price/Volume Stats
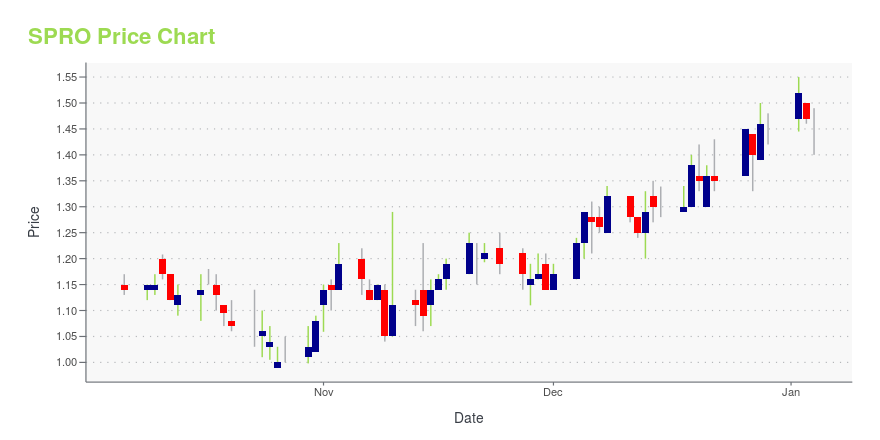
| Current price | $1.44 | 52-week high | $1.89 |
| Prev. close | $1.42 | 52-week low | $0.99 |
| Day low | $1.40 | Volume | 38,500 |
| Day high | $1.44 | Avg. volume | 226,896 |
| 50-day MA | $1.41 | Dividend yield | N/A |
| 200-day MA | $1.43 | Market Cap | 77.47M |
SPRO Stock Price Chart Interactive Chart >
Spero Therapeutics, Inc. (SPRO) Company Bio
Spero Therapeutics, Inc., a clinical-stage biopharmaceutical company, focuses on identifying, developing, and commercializing novel treatments for multi-drug resistant (MDR) bacterial infections in the United States. It is developing SPR994, an oral carbapenem-class antibiotic for use in adults to treat MDR gram-negative infections; SPR741 that has completed Phase I clinical trial to treat MDR gram-negative infections in the hospital setting; SPR206, an agent that is in preclinical development stage to disrupt the outer membrane of gram-negative bacteria; and SPR720, an oral antibiotic that is in preclinical development stage for the treatment of pulmonary non-tuberculous mycobacterial infections. The company was founded in 2013 and is based in Cambridge, Massachusetts.
Latest SPRO News From Around the Web
Below are the latest news stories about SPERO THERAPEUTICS INC that investors may wish to consider to help them evaluate SPRO as an investment opportunity.
Spero Therapeutics, Inc. (NASDAQ:SPRO) Q3 2023 Earnings Call TranscriptSpero Therapeutics, Inc. (NASDAQ:SPRO) Q3 2023 Earnings Call Transcript November 13, 2023 Spero Therapeutics, Inc. beats earnings expectations. Reported EPS is $0.04, expectations were $-0.15. Ted Jenkins: Thank you, operator, and thank you all for participating in today’s conference call. This afternoon, Spero Therapeutics released financial results and provided a pipeline update for the third […] |
Spero Therapeutics Announces Third Quarter 2023 Operating Results and Provides a Business UpdateSpecial Protocol Assessment Agreement with FDA for Phase 3 PIVOT-PO trial of tebipenem HBr; Trial expected to begin with First Patient, First Visit in 4Q 2023 Conference call and webcast at 4:30pm ET today CAMBRIDGE, Mass., Nov. 13, 2023 (GLOBE NEWSWIRE) -- Spero Therapeutics, Inc. (Nasdaq: SPRO), a multi-asset clinical-stage biopharmaceutical company, focused on identifying, developing, and commercializing treatments in high unmet need areas involving rare diseases and multi-drug resistant (MDR |
Spero Therapeutics to Provide Business Update and Report Third Quarter 2023 Financial Results on Monday, November 13, 2023CAMBRIDGE, Mass., Nov. 06, 2023 (GLOBE NEWSWIRE) -- Spero Therapeutics, Inc. (Nasdaq: SPRO), a multi-asset clinical-stage biopharmaceutical company, focused on identifying, developing and commercializing treatments in high unmet need areas involving rare diseases and multi-drug resistant (MDR) bacterial infections, today announced that it will host a conference call and live audio webcast on Monday, November 13, 2023 at 4:30 p.m. ET to report its third quarter 2023 financial results and provide |
Spero Therapeutics to Present at Upcoming November Investor ConferencesCAMBRIDGE, Mass., Nov. 01, 2023 (GLOBE NEWSWIRE) -- Spero Therapeutics, Inc. (Nasdaq: SPRO) (Spero), a multi-asset clinical-stage biopharmaceutical company, focused on identifying, developing and commercializing treatments in high unmet need areas involving rare diseases and multi-drug resistant (MDR) bacterial infections, today announced that Sath Shukla, President and Chief Executive Officer, will be available for one-on-one meetings at the Truist Securities BioPharma Symposium, to be held at |
Spero Therapeutics Appoints Esther Rajavelu as Chief Financial Officer and Chief Business OfficerMs. Rajavelu brings over two decades of experience in the life sciences sector focused on strategic growth, financing, M&A, and investor relations.CAMBRIDGE, Mass., Nov. 01, 2023 (GLOBE NEWSWIRE) -- Spero Therapeutics, Inc. (Nasdaq: SPRO), a multi-asset clinical-stage biopharmaceutical company, focused on identifying, developing, and commercializing treatments in high unmet need areas involving rare diseases and multi-drug resistant (MDR) bacterial infections, today announced the appointment of |
SPRO Price Returns
| 1-mo | 7.46% |
| 3-mo | -1.37% |
| 6-mo | -9.43% |
| 1-year | 1.41% |
| 3-year | -89.87% |
| 5-year | -85.64% |
| YTD | -2.04% |
| 2023 | -15.03% |
| 2022 | -89.19% |
| 2021 | -17.43% |
| 2020 | 101.66% |
| 2019 | 56.34% |
Continue Researching SPRO
Want to do more research on Spero Therapeutics Inc's stock and its price? Try the links below:Spero Therapeutics Inc (SPRO) Stock Price | Nasdaq
Spero Therapeutics Inc (SPRO) Stock Quote, History and News - Yahoo Finance
Spero Therapeutics Inc (SPRO) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...