Sorrento Therapeutics Inc. (SRNE): Price and Financial Metrics
SRNE Price/Volume Stats
| Current price | $0.01 | 52-week high | $0.35 |
| Prev. close | $0.01 | 52-week low | $0.00 |
| Day low | $0.01 | Volume | 49,400 |
| Day high | $0.01 | Avg. volume | 322,945 |
| 50-day MA | $0.01 | Dividend yield | N/A |
| 200-day MA | $0.04 | Market Cap | 5.24M |
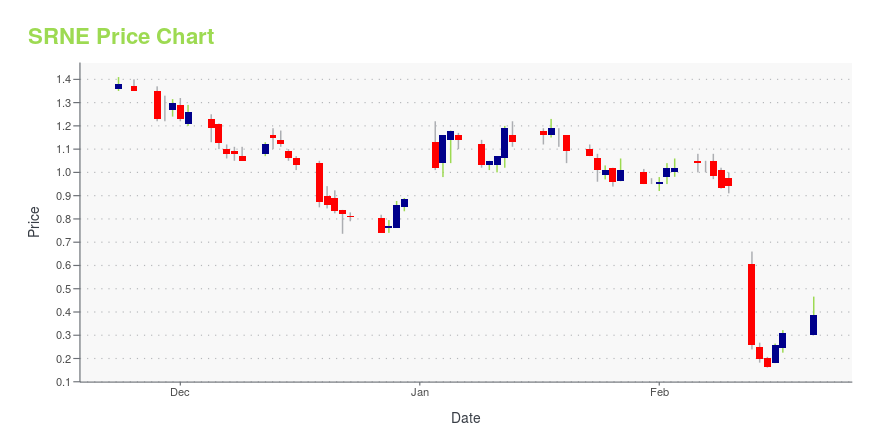
SRNE Stock Price Chart Interactive Chart >
Sorrento Therapeutics Inc. (SRNE) Company Bio
Sorrento Therapeutics, Inc. engages in the research, development and manufacture of biopharmaceutical products. It focuses on therapies to treat specific stages in the evolution of cancer, from elimination, to equilibrium and escape which include biosimilars, immuno-oncology antibodies, cellular therapy, cell internalizing antibodies, and antibody drug conjugates. The company was founded by Henry H. Ji in 2006 and is headquartered in San Diego, CA.
Latest SRNE News From Around the Web
Below are the latest news stories about SORRENTO THERAPEUTICS INC that investors may wish to consider to help them evaluate SRNE as an investment opportunity.
Scilex Holding Company announces the commercial launch of ELYXYBTM (celecoxib oral solution) in the U.S., strengthening its leadership position in non-opioid pain managementELYXYBTM is a first-line treatment and the only FDA-approved, ready-to-use oral solution for the acute treatment of migraine, with or without aura, in adults.1 The U.S. oral migraine drug market size is estimated to be $1.8 billion in 2022.2There is strong evidence for the use of NSAIDs as a first-line treatment for migraine. Celecoxib is in the same class of agents, is fast acting, and has the potential to have the lowest GI side effects of all NSAIDs.3Strong re-launch commercialization efforts |
The $75 Million Reason Sorrento (SRNE) Stock Is Moving TodaySRNE stock is moving this morning after soaring about 25% yesterday. |
Alphabet Layoffs 2023: What to Know About the Latest Google Job CutsAlphabet (GOOGL,GOOG) layoffs are worth checking on Wednesday as recent reports claim Google job cuts are coming to Ireland. |
Meta Platforms Layoffs 2023: Are More Facebook Job Cuts Coming?Meta Platforms (META) layoffs are a hot topic on Wednesday as reports spread that the Facebook parent company is planning more job cuts. |
Today’s Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on WednesdayWe're starting off the day with a breakdown of the biggest pre-market stock movers traders need to watch on Wednesday morning! |
SRNE Price Returns
| 1-mo | 0.00% |
| 3-mo | -27.01% |
| 6-mo | -64.29% |
| 1-year | -97.02% |
| 3-year | -99.88% |
| 5-year | -99.62% |
| YTD | -91.90% |
| 2023 | -86.06% |
| 2022 | -80.95% |
| 2021 | -31.87% |
| 2020 | 101.92% |
| 2019 | 40.83% |
Continue Researching SRNE
Want to do more research on Sorrento Therapeutics Inc's stock and its price? Try the links below:Sorrento Therapeutics Inc (SRNE) Stock Price | Nasdaq
Sorrento Therapeutics Inc (SRNE) Stock Quote, History and News - Yahoo Finance
Sorrento Therapeutics Inc (SRNE) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...