Stoke Therapeutics Inc. (STOK): Price and Financial Metrics
STOK Price/Volume Stats
| Current price | $15.86 | 52-week high | $17.58 |
| Prev. close | $14.56 | 52-week low | $3.35 |
| Day low | $14.79 | Volume | 942,900 |
| Day high | $15.97 | Avg. volume | 888,408 |
| 50-day MA | $14.54 | Dividend yield | N/A |
| 200-day MA | $8.66 | Market Cap | 826.64M |
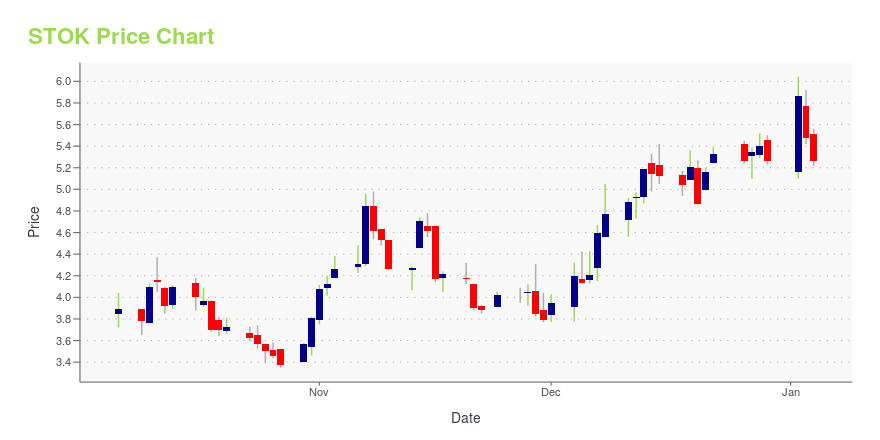
STOK Stock Price Chart Interactive Chart >
Stoke Therapeutics Inc. (STOK) Company Bio
Stoke Therapeutics, Inc. was founded in 2014 by Professor Adrian Krainer, PhD. and Isabel Aznarez, PhD to research and develop treatments for the underlying cause of genetic diseases. The company focuses on developing antisense oligonucleotide medicines that increase gene expression to treat genetic epilepsies and other severe monogenic diseases, including genetic conditions affecting the central nervous system, eye, liver, and kidney. Stoke’s proprietary platform, Targeted Augmentation of Nuclear Gene Output (TANGO) allows for the delivery of disease-modifying therapies in a precise and controlled manner. Edward M. Kaye, M.D., currently serves as the company’s director and Chief Executive Officer.
Latest STOK News From Around the Web
Below are the latest news stories about STOKE THERAPEUTICS INC that investors may wish to consider to help them evaluate STOK as an investment opportunity.
Wall Street Analysts Predict a 220.81% Upside in Stoke Therapeutics, Inc. (STOK): Here's What You Should KnowThe consensus price target hints at a 220.8% upside potential for Stoke Therapeutics, Inc. (STOK). While empirical research shows that this sought-after metric is hardly effective, an upward trend in earnings estimate revisions could mean that the stock will witness an upside in the near term. |
Is Stoke Therapeutics (NASDAQ:STOK) In A Good Position To Deliver On Growth Plans?We can readily understand why investors are attracted to unprofitable companies. For example, although Amazon.com made... |
Stoke Therapeutics Presents Data From Multiple Studies of Children and Adolescents With Dravet Syndrome at the American Epilepsy Society (AES) 2023 Annual MeetingBEDFORD, Mass., December 01, 2023--Stoke Therapeutics, Inc. (Nasdaq: STOK), a biotechnology company dedicated to addressing the underlying cause of severe diseases by upregulating protein expression with RNA-based medicines, today announced highlights from presentations of clinical data at the American Epilepsy Society (AES) 2023 Annual Meeting December 1 – 5, in Orlando, Florida. Together, these data support the company’s continued progress to develop STK-001 as the first disease-modifying medi |
Stoke Therapeutics to Present Data from the Company’s Dravet Syndrome Clinical Program at the American Epilepsy Society 2023 Annual MeetingBEDFORD, Mass., November 21, 2023--Stoke Therapeutics, Inc. (Nasdaq: STOK), a biotechnology company dedicated to addressing the underlying cause of severe diseases by upregulating protein expression with RNA-based medicines, today announced that presentations related to the Company’s work in Dravet syndrome will be presented at the American Epilepsy Society (AES) 2023 Annual Meeting, taking place December 1 – 5, in Orlando, Florida. The company is advancing STK-001 as potentially the first medic |
Stoke Therapeutics Inc (STOK) Reports Q3 Financial Results and Progress in Dravet Syndrome TreatmentKey Developments in STK-001 Studies and Financial Health Indicators |
STOK Price Returns
| 1-mo | 22.76% |
| 3-mo | 27.80% |
| 6-mo | 222.36% |
| 1-year | 158.31% |
| 3-year | -45.29% |
| 5-year | -33.61% |
| YTD | 201.52% |
| 2023 | -43.01% |
| 2022 | -61.53% |
| 2021 | -61.26% |
| 2020 | 118.68% |
| 2019 | N/A |

Loading social stream, please wait...