Synlogic, Inc. (SYBX): Price and Financial Metrics
SYBX Price/Volume Stats
| Current price | $1.67 | 52-week high | $8.85 |
| Prev. close | $1.68 | 52-week low | $1.35 |
| Day low | $1.59 | Volume | 1,895 |
| Day high | $1.68 | Avg. volume | 72,877 |
| 50-day MA | $1.57 | Dividend yield | N/A |
| 200-day MA | $2.20 | Market Cap | 19.55M |
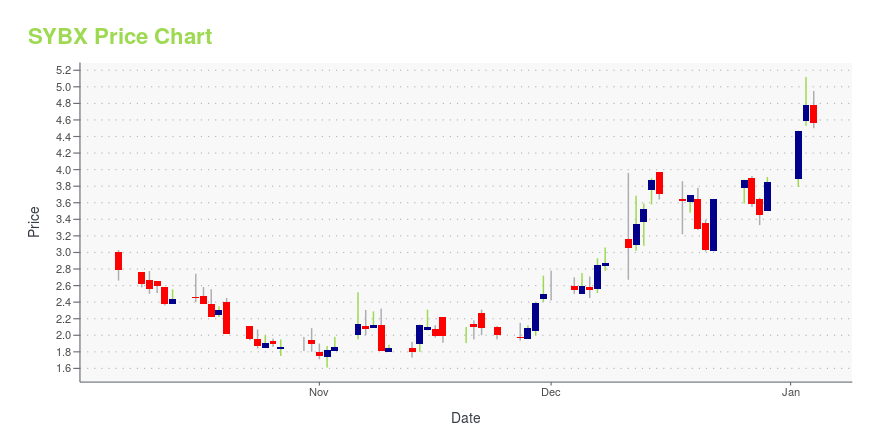
SYBX Stock Price Chart Interactive Chart >
Synlogic, Inc. (SYBX) Company Bio
Synlogic, Inc., a biopharmaceutical company, offers synthetic biotics that perform specific metabolic functions from the gut microbiome to correct missing or dysfunctional metabolic activities throughout the body. It provides SYNB1010 for urea cycle disorder, a synthetic biotic that is designed to remove excess ammonia from the blood; and SYNB2010 for phenylketonuria, a synthetic biotic that is designed to remove excess phenylalanine from the blood. The company was founded in 2013 and is based in Cambridge, Massachusetts.
Latest SYBX News From Around the Web
Below are the latest news stories about SYNLOGIC INC that investors may wish to consider to help them evaluate SYBX as an investment opportunity.
Synlogic Announces Contract with the Air Force Research Lab- Selection of Synlogic reflects Company’s leadership in process development and manufacturing of therapeutics based on synthetic biology -CAMBRIDGE, Mass., Dec. 27, 2023 (GLOBE NEWSWIRE) -- Synlogic, Inc. (Nasdaq: SYBX), a clinical-stage biotechnology company advancing novel, oral, non-systemically absorbed biotherapeutics to transform the care of serious diseases, announced that it has entered into an approximately $1 million subcontract under an Air Force Research Lab (AFRL) prime contract to |
Synlogic Announces Abstract Accepted for Poster Presentation at the International Conference on Microbiome Engineering 2023- Poster presentation on SYNB1353 program for homocystinuria includes findings from process improvements to improve activity of methionine degradation - CAMBRIDGE, Mass., Dec. 07, 2023 (GLOBE NEWSWIRE) -- Synlogic, Inc. (Nasdaq: SYBX), a clinical-stage biotechnology company advancing novel, oral, non-systemically absorbed biotherapeutics to transform the care of serious diseases, announced that an abstract outlining the SYNB1353 program as a potential treatment for homocystinuria (HCU), includin |
Sidoti Events, LLC’s Virtual November Micro-Cap ConferenceNEW YORK, NY / ACCESSWIRE / November 14, 2023 / Sidoti Events, LLC, an affiliate of Sidoti & Company, LLC, has released the presentation schedule and weblinks for its two-day November Micro-Cap Conference taking place Wednesday and Thursday, November ... |
Synlogic Inc (SYBX) Reports Q3 2023 Financial Results with Extended Cash Runway and Clinical ...Key Developments and Financial Highlights for Investors |
Synlogic Reports Third Quarter 2023 Financial Results and Provides Corporate Update- Underwritten public offering of $21.0 million and earned $2.5 million milestone payment from Roche have extended cash runway into the first half of 2025 - - Progress of the Synpheny-3 pivotal trial in phenylketonuria (PKU) supports enrollment completion in 2024, with top-line data in the first half of 2025 - - Company received Fast Track Designation from the FDA for labafenogene marselecobac (SYNB1934) as a potential treatment for PKU - CAMBRIDGE, Mass., Nov. 09, 2023 (GLOBE NEWSWIRE) -- Synlo |
SYBX Price Returns
| 1-mo | 9.87% |
| 3-mo | -10.70% |
| 6-mo | -53.61% |
| 1-year | -76.81% |
| 3-year | -96.53% |
| 5-year | -98.25% |
| YTD | -56.62% |
| 2023 | -66.23% |
| 2022 | -68.60% |
| 2021 | 12.04% |
| 2020 | -16.28% |
| 2019 | -63.20% |
Continue Researching SYBX
Want to do more research on Synlogic Inc's stock and its price? Try the links below:Synlogic Inc (SYBX) Stock Price | Nasdaq
Synlogic Inc (SYBX) Stock Quote, History and News - Yahoo Finance
Synlogic Inc (SYBX) Stock Price and Basic Information | MarketWatch

Loading social stream, please wait...