Verve Therapeutics, Inc. (VERV): Price and Financial Metrics
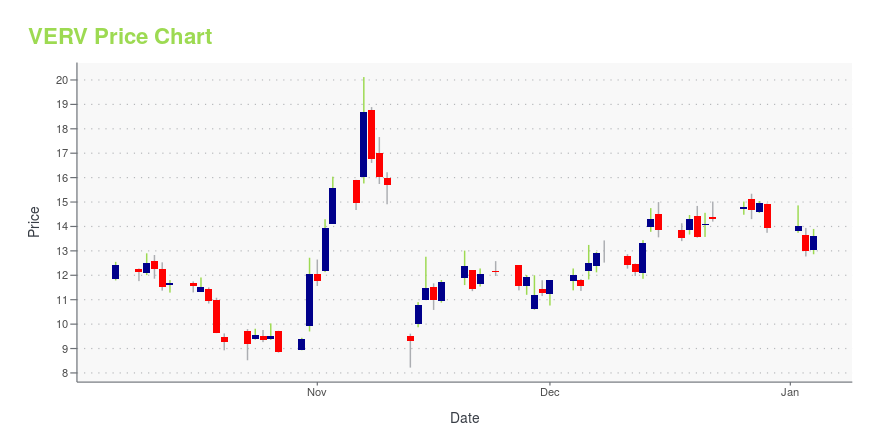
VERV Price/Volume Stats
| Current price | $7.40 | 52-week high | $20.72 |
| Prev. close | $7.48 | 52-week low | $4.59 |
| Day low | $7.27 | Volume | 857,799 |
| Day high | $7.90 | Avg. volume | 1,299,952 |
| 50-day MA | $5.82 | Dividend yield | N/A |
| 200-day MA | $10.19 | Market Cap | 621.34M |
VERV Stock Price Chart Interactive Chart >
Verve Therapeutics, Inc. (VERV) Company Bio
Verve Therapeutics, Inc. is a genetic medicines company engaged in the development of a new approach to the care of cardiovascular disease. It aims to transform treatment from chronic management to single-course gene editing medicines. Its goal is to disrupt the chronic care model for CVD by providing a new therapeutic approach with single-course in vivo gene editing treatments focused on addressing the root causes of this highly prevalent and life-threatening disease.
Latest VERV News From Around the Web
Below are the latest news stories about VERVE THERAPEUTICS INC that investors may wish to consider to help them evaluate VERV as an investment opportunity.
Wall Street Analysts Believe Verve Therapeutics (VERV) Could Rally 260.31%: Here's is How to TradeThe average of price targets set by Wall Street analysts indicates a potential upside of 260.3% in Verve Therapeutics (VERV). While the effectiveness of this highly sought-after metric is questionable, the positive trend in earnings estimate revisions might translate into an upside in the stock. |
Verve Therapeutics Announces Closing of Public Offering of Common Stock, Full Exercise by Underwriters of Option to Purchase Additional Shares and Closing of Concurrent Private PlacementBOSTON, Dec. 01, 2023 (GLOBE NEWSWIRE) -- Verve Therapeutics, Inc. (Nasdaq: VERV), a clinical-stage biotechnology company pioneering a new approach to the care of cardiovascular disease with single-course gene editing medicines, today announced the closing of its previously announced underwritten public offering of 14,375,000 shares of its common stock at a public offering price of $10.00 per share, which includes 1,875,000 shares issued upon the exercise in full by the underwriters of their opt |
Today’s Biggest Pre-Market Stock Movers: 10 Top Gainers and Losers on WednesdayPre-market stock movers are a good way to start the day as we dive into the biggest news worth checking out on Wednesday morning! |
Verve Therapeutics Announces Pricing of Public Offering of Common Stock and Concurrent Private PlacementBOSTON, Nov. 28, 2023 (GLOBE NEWSWIRE) -- Verve Therapeutics, Inc. (Nasdaq: VERV), a clinical-stage biotechnology company pioneering a new approach to the care of cardiovascular disease with single-course gene editing medicines, today announced the pricing of an underwritten public offering of 12,500,000 shares of its common stock at a public offering price of $10.00 per share, for total gross proceeds of $125.0 million, before deducting underwriting discounts and commissions and other offering |
Verve Therapeutics Announces Proposed Public Offering of Common Stock and Concurrent Private PlacementBOSTON, Nov. 28, 2023 (GLOBE NEWSWIRE) -- Verve Therapeutics, Inc. (Nasdaq: VERV), a clinical-stage biotechnology company pioneering a new approach to the care of cardiovascular disease with single-course gene editing medicines, today announced that it has commenced an underwritten public offering of shares of its common stock. All of the shares are being offered by Verve. In addition, Verve expects to grant the underwriters a 30-day option to purchase up to an additional 15% of the shares of it |
VERV Price Returns
| 1-mo | 47.41% |
| 3-mo | 20.13% |
| 6-mo | -35.88% |
| 1-year | -61.97% |
| 3-year | -85.18% |
| 5-year | N/A |
| YTD | -46.92% |
| 2023 | -27.96% |
| 2022 | -47.52% |
| 2021 | N/A |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...