Verrica Pharmaceuticals Inc. (VRCA): Price and Financial Metrics
VRCA Price/Volume Stats
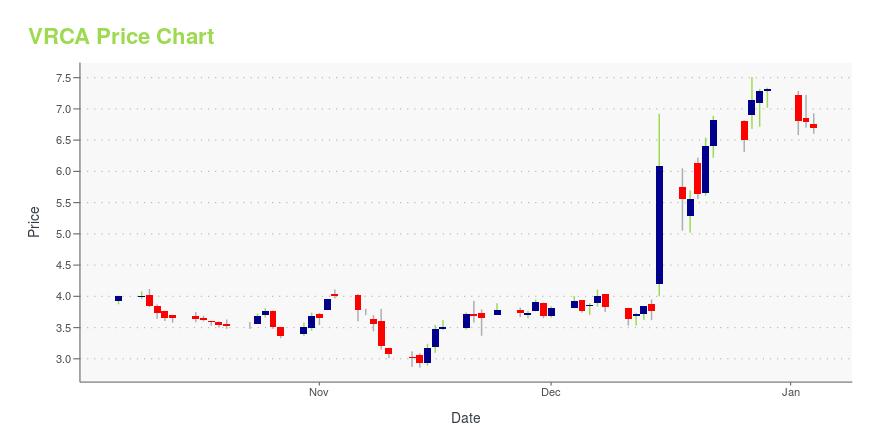
| Current price | $7.24 | 52-week high | $11.41 |
| Prev. close | $7.25 | 52-week low | $2.86 |
| Day low | $7.01 | Volume | 157,422 |
| Day high | $7.34 | Avg. volume | 192,680 |
| 50-day MA | $8.06 | Dividend yield | N/A |
| 200-day MA | $6.01 | Market Cap | 307.12M |
VRCA Stock Price Chart Interactive Chart >
Verrica Pharmaceuticals Inc. (VRCA) Company Bio
Verrica Pharmaceuticals Inc. develops and commercializes dermatological treatments in the United States. The company is advancing its lead product VP-102, a proprietary topical therapy, in various common skin indications, including Molluscum contagiosum and Verruca Vulgaris. It is also developing 2 Phase 3 trials in molluscum contagiosum under NCT03377803 and NCT03377790. The company is based in West Chester, Pennsylvania.
Latest VRCA News From Around the Web
Below are the latest news stories about VERRICA PHARMACEUTICALS INC that investors may wish to consider to help them evaluate VRCA as an investment opportunity.
Despite recent sales, Verrica Pharmaceuticals Inc. (NASDAQ:VRCA) insiders remain the largest stockholders with 39% ownershipKey Insights Verrica Pharmaceuticals' significant insider ownership suggests inherent interests in company's expansion... |
Why Is Verrica Pharmaceuticals (VRCA) Stock Up 40% Today?With Verrica Pharmaceuticals releasing positive Phase 3 trial results for a skin disease therapeutic, VRCA stock skyrocketed in response. |
Verrica Pharmaceuticals’ Development and Commercialization Partner, Torii Pharmaceutical Co., Ltd., Announces Positive Top-line Results from a Confirmatory Phase 3 Trial of TO-208 for the Treatment of Molluscum Contagiosum in JapanWEST CHESTER, Pa., Dec. 15, 2023 (GLOBE NEWSWIRE) -- Verrica Pharmaceuticals Inc. (“Verrica” or “the Company”) (Nasdaq: VRCA), a dermatology therapeutics company developing medications for skin diseases requiring medical interventions, today announced that its development and commercialization partner, Torii Pharmaceutical Co., Ltd. (“Torii”), reported positive top-line results from its Phase 3 trial of TO-208 (referred to as VP-102 and marketed as YCANTH™ in the U.S.) for the treatment of Mollu |
Verrica Pharmaceuticals Inc. (NASDAQ:VRCA) Q3 2023 Earnings Call TranscriptVerrica Pharmaceuticals Inc. (NASDAQ:VRCA) Q3 2023 Earnings Call Transcript November 9, 2023 Verrica Pharmaceuticals Inc. misses on earnings expectations. Reported EPS is $-0.54 EPS, expectations were $-0.22. Operator: Good morning, ladies and gentlemen, and welcome to the Verrica Pharmaceuticals Third Quarter 2023 Corporate Update and Earnings Conference Call. As a reminder, this conference is being […] |
Verrica Pharmaceuticals Announces Presentation at the Jefferies Global Healthcare Conference in LondonWEST CHESTER, Pa., Nov. 10, 2023 (GLOBE NEWSWIRE) -- Verrica Pharmaceuticals Inc. (“Verrica” or “the Company”) (Nasdaq: VRCA), a dermatology therapeutics company developing medications for skin diseases requiring medical interventions, today announced Ted White, Verrica President & Chief Executive Officer, will present at the upcoming Jefferies Global Healthcare Conference, which is being held in London from November 14-16, 2023. Event details:Date: Wednesday, November 15, 2023Time: 2:00 pm GMTL |
VRCA Price Returns
| 1-mo | 2.70% |
| 3-mo | 5.39% |
| 6-mo | 25.04% |
| 1-year | 39.77% |
| 3-year | -32.90% |
| 5-year | -33.64% |
| YTD | -1.09% |
| 2023 | 166.18% |
| 2022 | -69.98% |
| 2021 | -20.42% |
| 2020 | -27.56% |
| 2019 | 94.97% |

Loading social stream, please wait...