4D Molecular Therapeutics Inc. (FDMT): Price and Financial Metrics
FDMT Price/Volume Stats
| Current price | $23.25 | 52-week high | $36.25 |
| Prev. close | $23.53 | 52-week low | $9.44 |
| Day low | $22.75 | Volume | 322,774 |
| Day high | $23.86 | Avg. volume | 1,032,306 |
| 50-day MA | $28.51 | Dividend yield | N/A |
| 200-day MA | $19.19 | Market Cap | 1.19B |
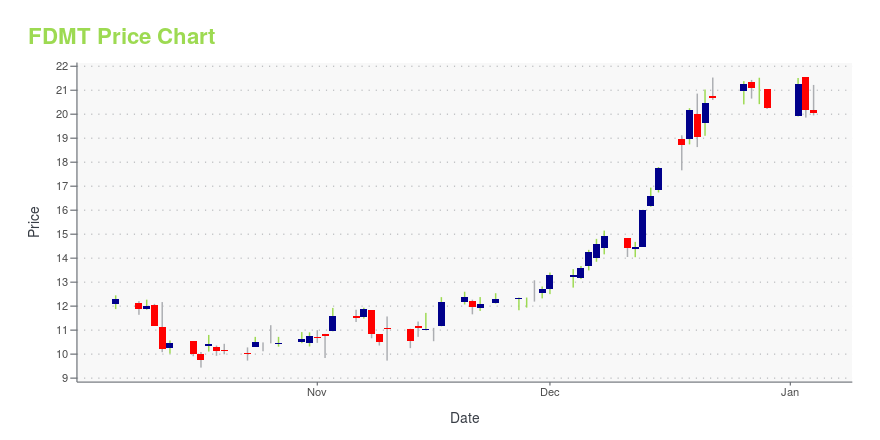
FDMT Stock Price Chart Interactive Chart >
4D Molecular Therapeutics Inc. (FDMT) Company Bio
4D Molecular Therapeutics is a biotechnology company. The Company designs, develops, and commercializes transformative gene therapeutic products for unmet medical conditions.
Latest FDMT News From Around the Web
Below are the latest news stories about 4D MOLECULAR THERAPEUTICS INC that investors may wish to consider to help them evaluate FDMT as an investment opportunity.
4DMT Receives FDA Regenerative Medicine Advanced Therapy (RMAT) Designation for 4D-150 Genetic Medicine for Intravitreal Treatment of Wet AMD, the First RMAT Designation in Wet AMDDesignation follows interim Phase 1 PRISM clinical data for 4D-150 that demonstrated an encouraging safety, tolerability and clinical activity profile in patients with wet age-related macular degeneration (wet AMD) 4D-150 is the first therapeutic candidate to receive either RMAT or Breakthrough Designation for wet AMDRMAT designation follows Priority Medicines (PRIME) designation received from the European Medicines Agency (EMA) in October 2023Receiving both RMAT and PRIME enables increased coll |
4DMT to Present Interim 24 Week Data from Randomized Phase 2 PRISM Clinical Trial of 4D-150 in High Treatment Need Wet AMD Patients at Angiogenesis, Exudation, and Degeneration 2024 ConferenceInitial interim landmark data analysis (N=50 at 24 Weeks) to be presented by Arshad M. Khanani, M.D., M.A., FASRS, Principal Investigator of the PRISM clinical trial, on Saturday, February 3, 2024 at 4:20 p.m. ET EMERYVILLE, Calif., Dec. 04, 2023 (GLOBE NEWSWIRE) -- 4D Molecular Therapeutics (Nasdaq: FDMT, 4DMT or the Company), a clinical-stage biotherapeutics company with three novel, highly targeted next generation AAV vectors currently in the clinic, today announced that the initial interim l |
4DMT to Participate in Upcoming Investor ConferencesEMERYVILLE, Calif., Nov. 15, 2023 (GLOBE NEWSWIRE) -- 4D Molecular Therapeutics, Inc. (Nasdaq: FDMT, 4DMT, or the Company), a genetic medicines company with three novel, highly targeted next generation AAV vectors currently in human clinical studies, announced today that management will participate in a fireside chat at an upcoming investor conference in November. Members of the management team will also be available for one-on-one meetings in connection with this meeting. 6th Annual Evercore IS |
Broker Revenue Forecasts For 4D Molecular Therapeutics, Inc. (NASDAQ:FDMT) Are Surging HigherShareholders in 4D Molecular Therapeutics, Inc. ( NASDAQ:FDMT ) may be thrilled to learn that the analysts have just... |
4D Molecular Therapeutics, Inc. (NASDAQ:FDMT) Consensus Forecasts Have Become A Little Darker Since Its Latest ReportA week ago, 4D Molecular Therapeutics, Inc. ( NASDAQ:FDMT ) came out with a strong set of third-quarter numbers that... |
FDMT Price Returns
| 1-mo | -35.18% |
| 3-mo | 32.86% |
| 6-mo | 122.06% |
| 1-year | 40.91% |
| 3-year | -38.49% |
| 5-year | N/A |
| YTD | 14.76% |
| 2023 | -8.78% |
| 2022 | 1.23% |
| 2021 | -47.07% |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...