Taysha Gene Therapies Inc. (TSHA): Price and Financial Metrics
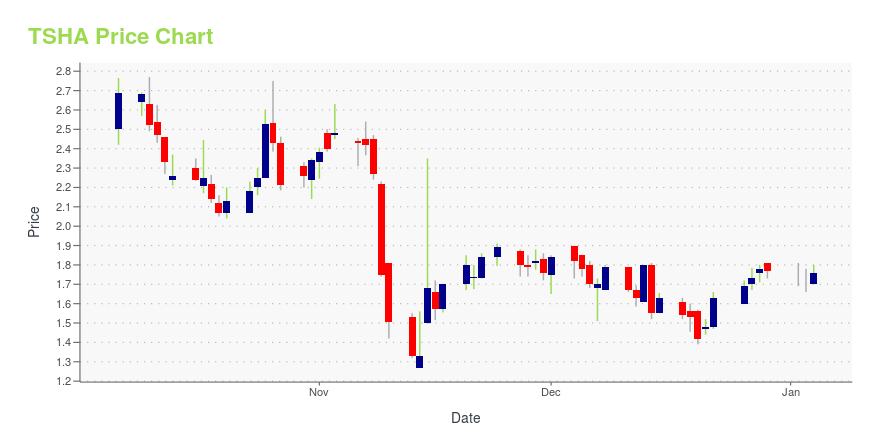
TSHA Price/Volume Stats
| Current price | $2.39 | 52-week high | $3.89 |
| Prev. close | $2.30 | 52-week low | $0.50 |
| Day low | $2.30 | Volume | 890,189 |
| Day high | $2.45 | Avg. volume | 2,570,028 |
| 50-day MA | $2.61 | Dividend yield | N/A |
| 200-day MA | $2.09 | Market Cap | 446.97M |
TSHA Stock Price Chart Interactive Chart >
Taysha Gene Therapies Inc. (TSHA) Company Bio
Taysha Gene Therapies, Inc. engages in the development and commercialization of adeno-associated viruses (AAV) based gene therapies for the treatment of monogenic diseases of the central nervous system. It also develops multiple gene therapy platforms which include AAV9 Discovery, Novel Capsid, and AAV Redosing. The company was founded by Steven Gray, Berge Minassian, and R. A. Session II on September 20, 2019 and is headquartered in Dallas, TX.
Latest TSHA News From Around the Web
Below are the latest news stories about TAYSHA GENE THERAPIES INC that investors may wish to consider to help them evaluate TSHA as an investment opportunity.
Trinity Capital Inc. Provides $40 Million Term Loan to Taysha Gene TherapiesTrinity Capital Inc. (NASDAQ: TRIN) ("Trinity"), a leading provider of diversified financial solutions to growth-stage companies, today announced the commitment of $40 million in term loans to Taysha Gene Therapies, Inc. (NASDAQ: TSHA) ("Taysha"), a clinical-stage gene therapy company, pursuant to a Loan and Security Agreement dated November 13, 2023, by and among Taysha, the lenders party thereto from time to time (the "Lenders"), and Trinity, as administrative agent and collateral agent for th |
Taysha Gene Therapies Announces Expanded Eligibility in REVEAL Phase 1/2 Adult Trial to Include Adolescent Rett Syndrome PatientsHealth Canada authorized the Company’s protocol amendment that expands eligibility to include patients aged 12 and older with stage four Rett syndrome in the REVEAL Phase 1/2 adult trial in Canada Protocol amendment broadens TSHA-102 treatment potential to both adolescent and adult patients with Rett syndrome Dosing of the third patient in the REVEAL Phase 1/2 adult trial (age 12+ protocol) and completion of cohort one (low dose) expected in the fourth quarter of 2023/first quarter of 2024 DALLA |
Insider Buying: Taysha Gene Therapies Board Observer Bought US$163k Of SharesInvestors who take an interest in Taysha Gene Therapies, Inc. ( NASDAQ:TSHA ) should definitely note that the Board... |
Party Time: Brokers Just Made Major Increases To Their Taysha Gene Therapies, Inc. (NASDAQ:TSHA) Earnings ForecastsCelebrations may be in order for Taysha Gene Therapies, Inc. ( NASDAQ:TSHA ) shareholders, with the analysts delivering... |
Analysts Are Upgrading Taysha Gene Therapies, Inc. (NASDAQ:TSHA) After Its Latest ResultsIt's been a good week for Taysha Gene Therapies, Inc. ( NASDAQ:TSHA ) shareholders, because the company has just... |
TSHA Price Returns
| 1-mo | -24.61% |
| 3-mo | 61.49% |
| 6-mo | 7.90% |
| 1-year | 258.05% |
| 3-year | -90.16% |
| 5-year | N/A |
| YTD | 35.03% |
| 2023 | -21.68% |
| 2022 | -80.60% |
| 2021 | -56.10% |
| 2020 | N/A |
| 2019 | N/A |

Loading social stream, please wait...